Alzheimer’s disease (AD) is the most common cause of dementia in the elderly. Amyloid-β plaques and neurofibrillary tangles (NFTs) are the main neuropathological hallmarks of AD, however it is becoming increasingly apparent that neuroinflammation plays a significant role in its pathophysiology.
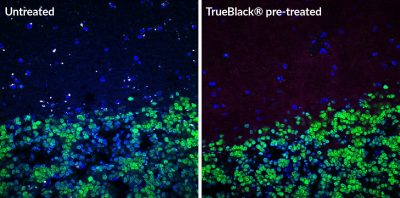
In a recent publication Dominy et al. elucidate the potential role of the oral pathogen Porphyromonas gingivalis, responsible for chronic periodontitis, an infection of the gums, in neurodegeneration and AD pathology. The authors demonstrate the presence of P. gingivalis in AD brains using a combination of immunoprecipitation, western blotting, and PCR. P. gingivalis DNA was also detected in the CSF of clinical AD patients suggesting that it may serve as a useful diagnostic marker for the disease. In addition, toxic proteases from the bacterium called gingipains, hypothesized to be involved in pathogenesis, were identified in AD hippocampal brain sections by immunofluorescence staining using TrueBlack® Lipofuscin Autofluorescence Quencher to eliminate tissue autofluorescence. Gingipain load correlated with levels of tau and ubiquitin in NFTs of AD brain plaques. Furthermore, the authors showed a dose-dependent effect of gingipain expression on tau protein fragmentation and neurotoxicity both in vivo and in vitro suggesting gingipain inhibition as a valuable treatment option. Small-molecule gingipain inhibitors were developed and their efficacy tested in a mouse model for chronic P. gingivalis infection.