Content #1
Content #1
Content #1
Cancer is a leading cause of death worldwide, with a reported 1.7 million new cancer cases within the United States in 2019 alone. When cancer is suspected, a tissue biopsy is often the first step toward a diagnosis. This procedure, in which cells are collected for examination, remains the gold standard for cancer diagnosis. A tissue biopsy allows healthcare providers to determine if cancer is present, shows what type of cancer it is, and gives clues about the patient’s prognosis. Unfortunately, it’s often invasive, risky, costly, and painful. These factors make repeated biopsies difficult and an impractical method for tracking tumors over time.
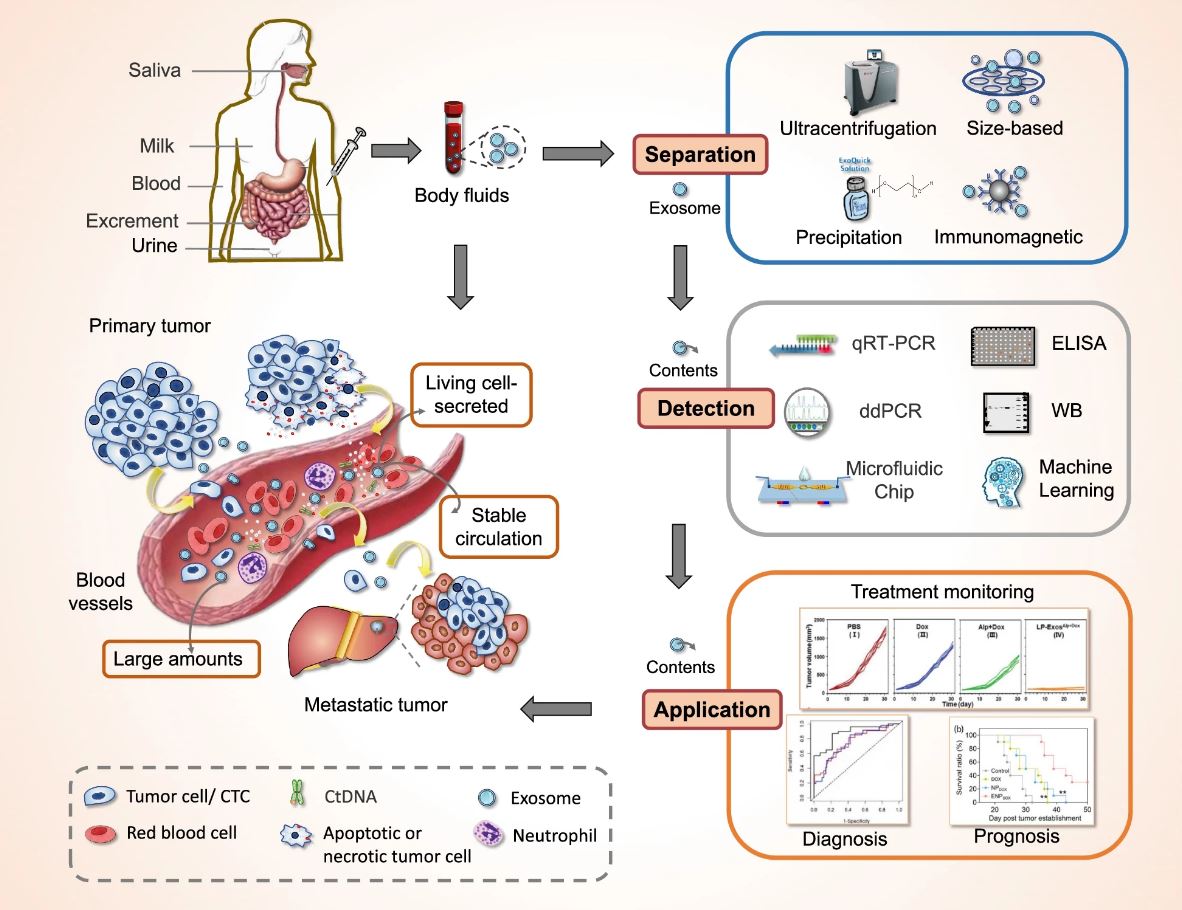
Overview of exosomes as a new target for liquid biopsy. Figure from Yu et al. 2022.1 Reproduced under the Creative Commons license.
These tiny EV’s were originally thought to be only vehicles for the disposal of cellular waste products, but exosomes are now being recognized for their important role in intercellular communication. Growing evidence suggests this is done through a variety of functional biomolecules, including nucleic acids, proteins, and lipids that exosomes transport. Now, researchers and clinicians are exploring whether the information carried by exosomes from their cells of origin might be useful for diagnosing and monitoring diseases. Tumor cell-derived exosomes, for example, have been shown to have immunomodulatory functions and play a role in signaling, direction, and invasion of metastatic cancer cells.1, 4-5
In addition to the informative cargo they transport, exosomes also display an array of other properties that make them well suited for disease diagnosis and monitoring. The following properties of exosomes make them attractive liquid biopsy candidates:
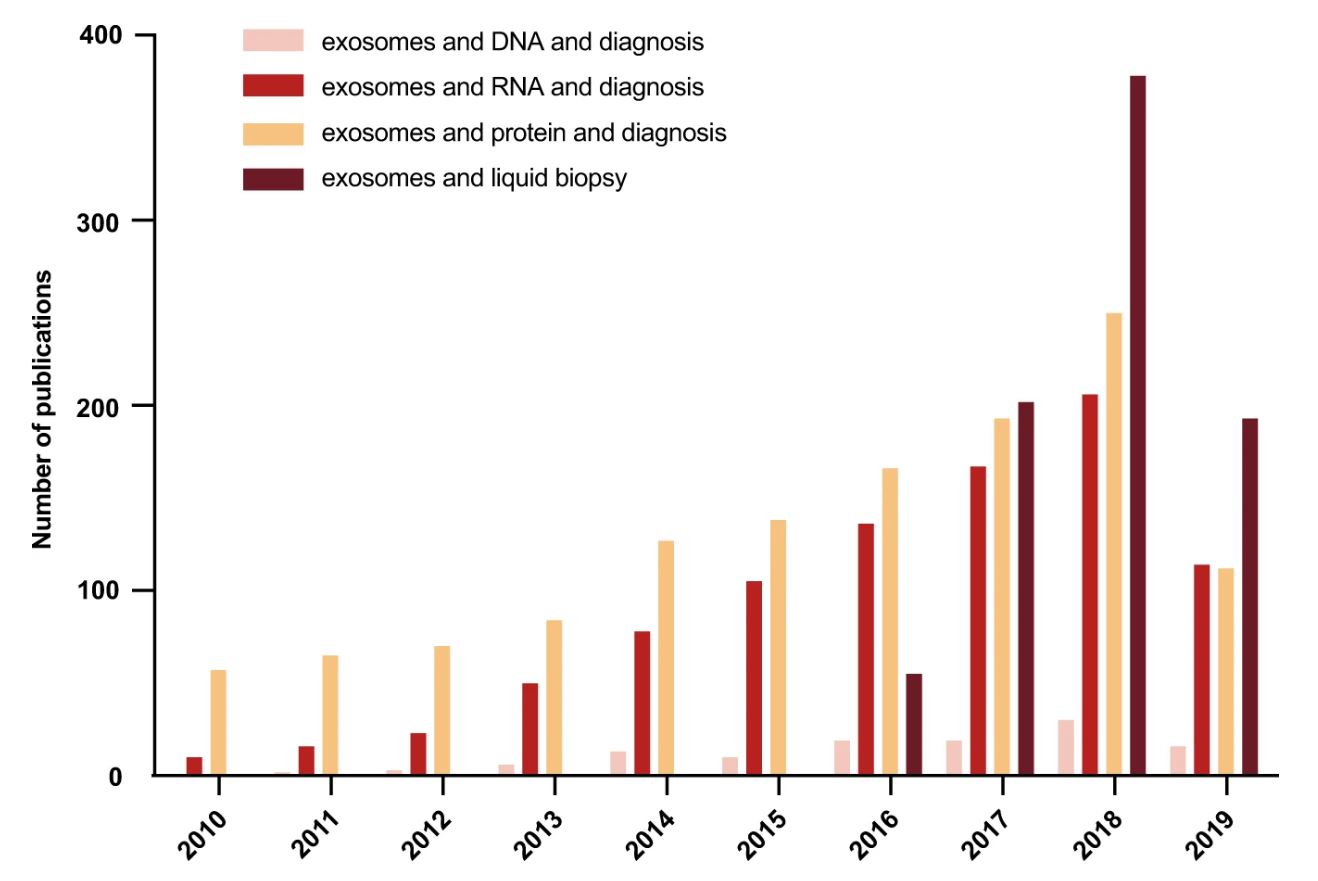
Publication frequencies of studies investigating different contents of exosomes in liquid biopsies for disease diagnosis over 10 years based on PubMed searches in January 2020. The number of studies in this new and fast moving field continues to grow. Figure from Zhou et al. 2020.4 Reproduced under the Creative Commons license.
Researchers have been hard at work over the past decade to validate the use of exosomes for liquid biopsies and mitigate the challenges associated with this technique. One of the main hurdles is effective, high-purity isolation due to their small size and heterogeneity in biofluids. For example, cancerous exosomes represent only a small percentage of exosomes in body fluid samples, which increases the requirements for ultrasensitive and specific detection. To solve these challenges, some researchers are developing methods to purify sub-populations of exosomes using immuno-capture with tissue-specific or cancer-specific antibodies.1, 4
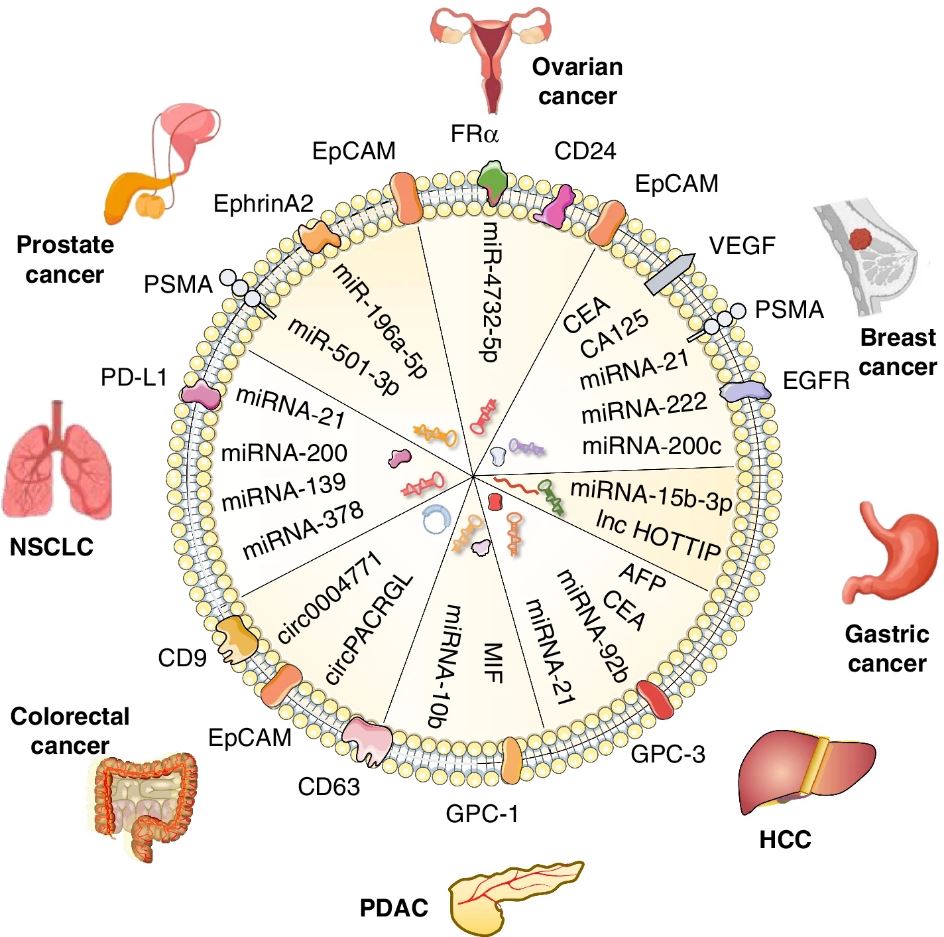
Cancer-derived exosomes are enriched in differentially expressed proteins and nucleic acids, which may act as biomarkers for the early diagnosis, stage classification, and prognosis prediction of different cancers. Figure from Yu et al. 2022.1 Reproduced under the Creative Commons license.
Early screening and accurate diagnosis are known to be the primary factors in reducing mortality and increasing the recovery rate for patients with tumors or precancerous lesions. Exosomes hold great potential as possible diagnostic and prognostic biomarkers and have been investigated in a variety of cancers. In addition, exosomes are involved in metastasis, and tumor immune suppression, suggesting that they might one day play a role in disease treatment as well. In fact, some clinical trials have already begun to explore the efficacy of using exosomes as therapeutics. 1, 3

Julianne Davis earned an MSc in Behavioral Neuroscience from the University of Washington, Seattle, where she examined the role of memory in cost-based decision-making. She has also studied sensory integration at the Allen Institute and the neural basis of feeding, thirst, and metabolism at the University of California, San Francisco as a research scientist. Currently, she is a Technical Writer and Support Scientist at Biotium.