Introduction
Flow cytometry is a prolific and powerful analytical tool used for biological research and the clinic. At its core, flow cytometry enables researchers to obtain highly specific information on individual cells within a sample. Recent advancements in this field have led to the development of spectral flow cytometry, a rapidly growing technology with significantly enhanced multiplexing capabilities over conventional flow cytometry. Several companies have capitalized on spectral flow with the release of the Sony ID7000™ and the well-known Cytek® Aurora. Where conventional flow cytometer instruments can detect panels with more than a dozen fluorophores, these new spectral flow cytometers are capable of accommodating multiparametric panels with upwards of 30 different fluorophores.
How Does Spectral Flow Cytometry Work?
Spectral flow cytometry shares much of the same hardware associated with conventional flow cytometry. Both systems employ standard fluidic and laser technologies that enable cell-by-cell spectral analysis. For both spectral and conventional flow cytometry methods, the process begins by delivering a sample stream through a flow chamber where cells move in a single file at a constant velocity, a process known as hydrodynamic focusing. This allows uniform and efficient excitation by a set of monochromatic lasers. Emitted photons are then detected and analyzed by a combination of optics and software to identify unique spectral emissions from fluorophore labeled biomolecules.
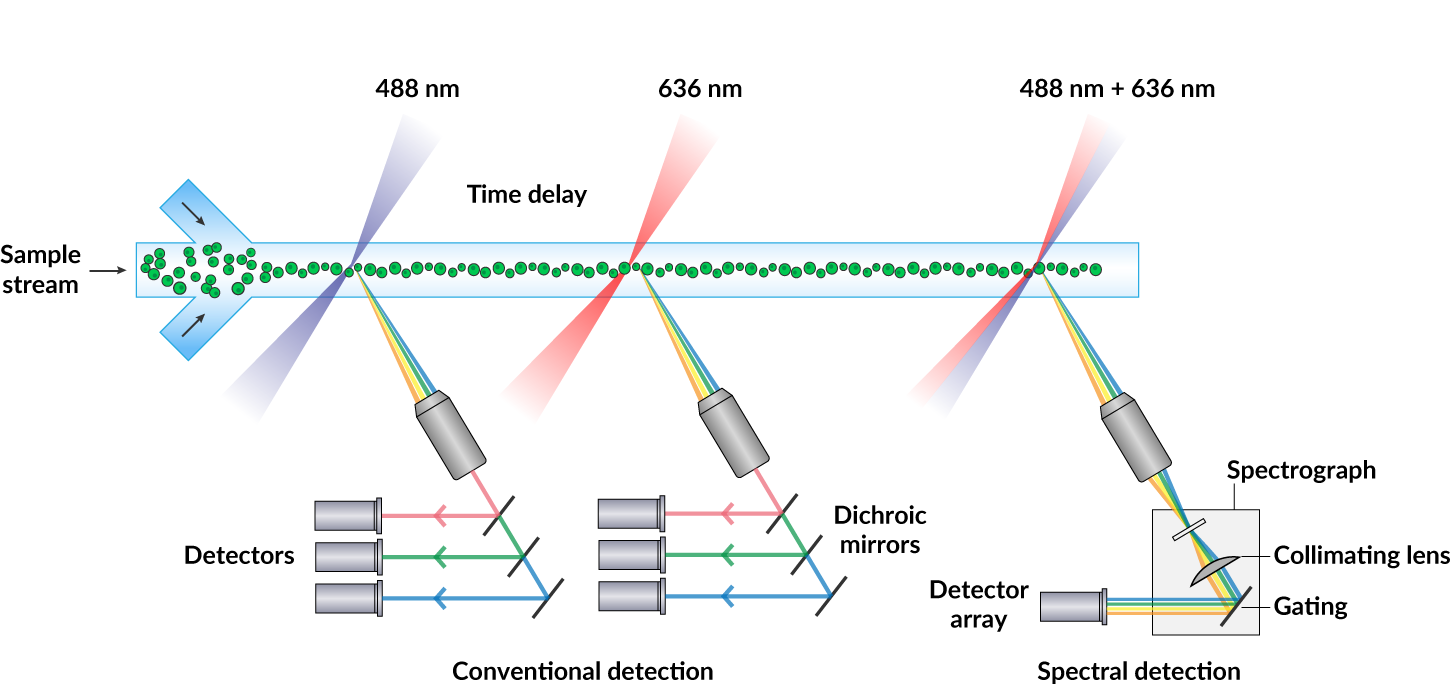
Capturing the Full Spectrum
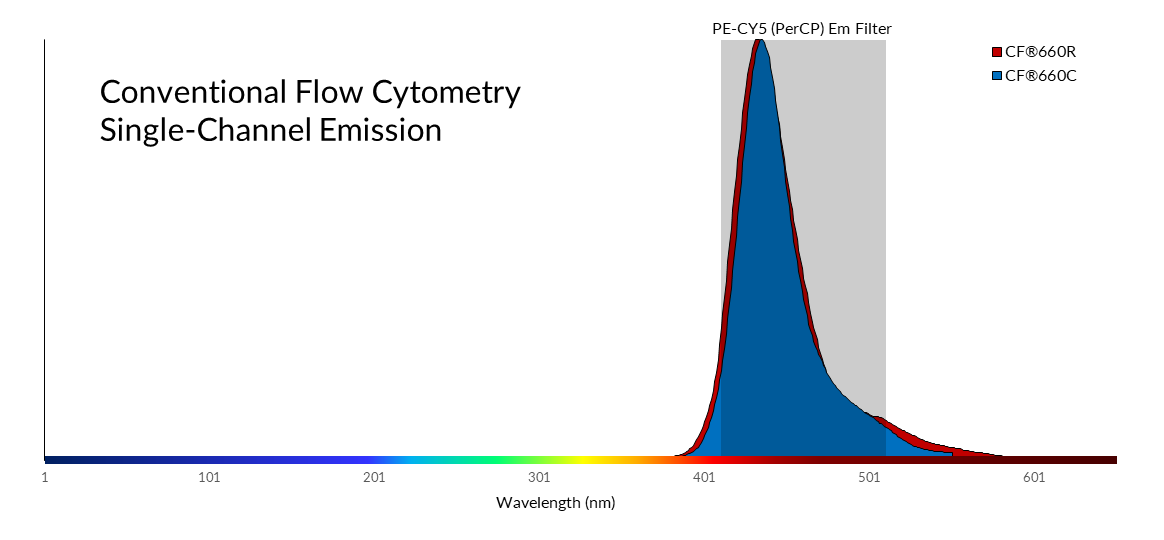
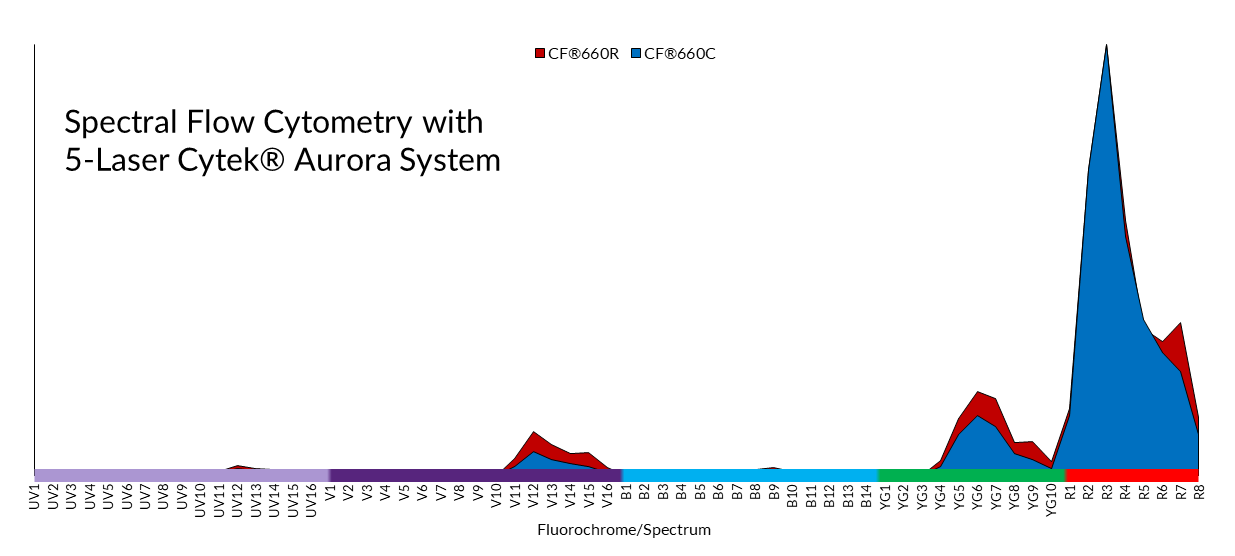
Where spectral flow differs most significantly from conventional flow cytometry is in the optical configuration and software analysis. In conventional flow cytometry, emitted photons are funneled through a series of dichroic mirrors and band pass filters that partition the light into a narrow bandwidth for detection by a set of photomultiplier tubes (PMTs). In contrast, spectral flow uses dispersive optics, such as prisms or spectrographs, that disperse photons according to wavelength across an array of detectors. This method broadens a fluorophore’s spectral profile by capturing the entire visible and near-IR spectrum. This allows for higher resolution spectral analysis over conventional optic configurations which detect only a small portion of the emission spectra. Consequently, fluorophores with very similar emission spectra that were once impossible to separate by conventional flow cytometry methods (Fig. 1), can now be distinguished through spectral flow cytometry. Figure 3 shows how the nearly identical spectral profiles of CF®660C and CF®660R can be distinguished with spectral flow cytometry, but not with conventional flow cytometry (Fig. 2). However, it should be noted that pairing of very spectrally similar dyes like CF®660C and CF®660R is not optimal dye pairing practice and would not be recommended even for spectral flow.
Compensation Vs. Spectral Unmixing
The same flow cytometry principles of panel design and spectral spillover are applicable to spectral flow cytometry. Designing multicolor fluorescence panels requires careful consideration of fluorophores with distinct spectral profiles that can be separated by the instrument. In conventional flow, when a multiparameter panel with a dozen or more fluorophores are used, overlap of emission spectra is unavoidable. This leads to noise in channels from emissions of unintended fluorophores, an issue known as spectral spillover. To address this, single-color control samples are run with each experiment to determine the amount of spectral overlap present in each detector. In conventional flow cytometry, a mathematical method known as compensation applies these controls to subtract the overlapping spectra and isolate a fluorophore’s emission profile. This is done through a correction factor that is derived from the ratio of intended and non-intended fluorophores emitting into a channel.
In spectral flow cytometry, more complex mathematical models are required to deal with spectral spillover. This is because the instrument must distinguish between multiple fluorescent profiles across the entire visible spectrum, rather than from a few distinct channels. The process of deconvoluting fluorophore emission spectra across an array of detectors is known as spectral unmixing. This type of compensation also requires experimental reference controls in addition to noise reducing mathematical algorithms such as the least squares method. This is particularly useful for cell culture samples prone to high autofluorescence. If you are interested in learning more about spectral flow cytometry, Nolan et. al have published a helpful review in Current Protocols in Cytometry.
| Conventional Flow Cytometry | Spectral Flow Cytometry | |
|---|---|---|
| Optics | Dichroic mirrors/band pass filters | Dispersive optics: prisms or spectrographs |
| Multiplexing | Limited by number of detectors and filters as well as emission overlap of fluorophores | Improved spectral resolution allows for more fluorophore probes per experiment |
| Spectral Resolution | Acquires only a narrow emission bandwidth from a single-laser excitation | Acquires the entire fluorophore spectral profile for each laser |
| Fluorophore Separation | Compensation | Spectral unmixing |
Take Full Advantage of Spectral Flow with CF® Dyes
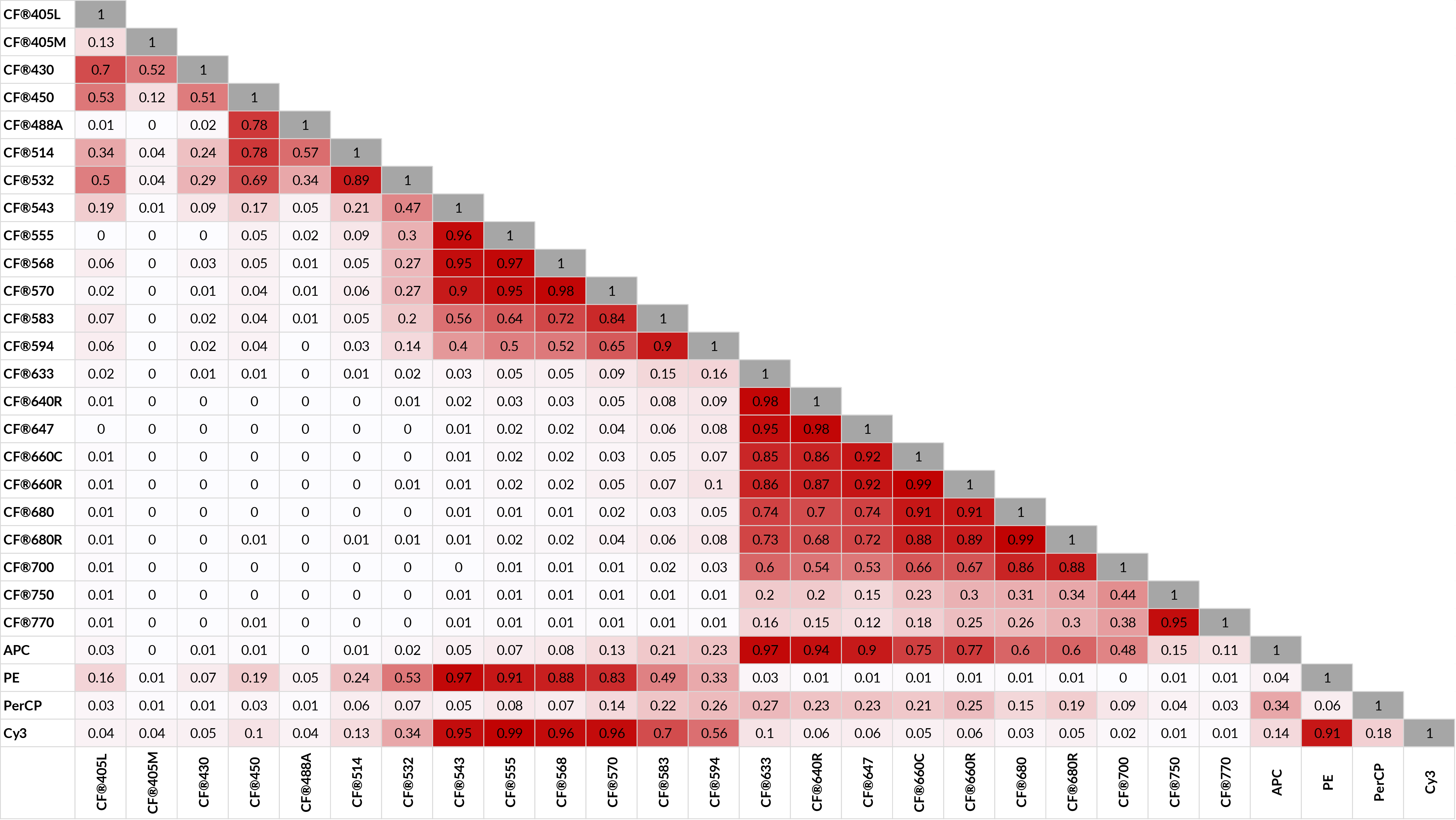
Biotium’s CF® Dyes allows you to maximize the multiplexing advantages of spectral flow by offering the largest selection of spectrally unique fluorophores, many of which are licensed by Cytek® Biosciences for use in spectral flow panels. See Figure 4 below for a similarity matrix of CF® Dyes that have been validated in spectral flow by Cytek®. In addition, CF® Dyes demonstrate superior brightness, solubility, and exceptional signal-to-noise compared to other fluorescent probes.
Validated & Published on the Cytek® Aurora
In a study published by Cytek in collaboration with Biotium, researchers acknowledge the limited number of spectrally unique fluorophores presents a challenge when designing panels that take full advantage of spectral flow cytometry technology. To overcome this challenge the study focuses on developing new flow cytometry reagents using CF® Dyes to accommodate the enhanced multiplexing capabilities of spectral flow cytometry. The authors conclude the addition of spectrally unique CF® Dyes enable support of high dimensional flow cytometry analysis of 30-40 fluorescent colors, a record in flow cytometry.

Link to the publication here.
Explore High-Performance CF® Dye Antibodies & Bioconjugates
We have a growing collection of over 2000 monoclonal CF® Dye primary and secondary antibody conjugates, as well as other bioconjugates. Our Biotium Choice line of primary antibodies are developed and optimized specifically for flow cytometry and are available with a selection of widely published clones against common targets.
Also see our Mix-n-Stain™ CF® Dye Antibody Labeling Kits for rapid and efficient conjugation to one of our CF® Dyes in as little as 15 minutes. For cell viability, please view our bright and extremely stable Live-or-Dye™ Fixable Viability Stains available in 16 different colors. Live-or-Dye™ stains 510/550, 665/685, 375/600, and 615/740 were designed specifically for spectral flow.
View our flow cytometry technology page to see a full listing of probes for flow cytometry and related products.

CF® Dye Similarity Index
Associated products
Live-or-Dye™ Fixable Viability Staining Kits
32022, 32022-T, 32021, 32021-T, 32016, 32017, 32018-T, 32002-T, 32014-T, 32003-T, 32009-T, 32004-T, 32012-T, 32015-T, 32005-T, 32007-T, 32006-T, 32013-T, 32008-T, 32011-T, 32018, 32002, 32014, 32003, 32009, 32004, 32012, 32005, 32006, 32015, 32007, 32013, 32008, - 32011View allHide
ViaFluor® SE Cell Proliferation Kits
30139, 30139-T, 30068-T, 30068, 30086-T, 30086, - 30050View allHide
NucView® Caspase-3 Enzyme Substrates
10405-T, 10405, 10407-T, 10407, 10402-T, 10402, 10403-T, 10403, 10406-T, 10406, 10408-T, - 10408View allHide
Dual Apoptosis Assay with NucView® 488 Caspase-3 Substrate & Annexin V
30030, 30067, - 30073View allHide
MitoView™ Mitochondrial Dyes
70070-T, 70070, 70054-T, 70054, 70055-T, 70055, 70075-50ug, 70075, 70068-T, - 70068View allHide
Flow Cytometry Fixation/Permeabilization Kit
NucSpot® Far-Red, 1000X in DMSO
40085, - 40085-TView allHide
Mix-n-Stain™ CF® Dye Antibody Labeling Kits
92576, 92577, 92578, 92582, 92583, 92584, 92579, 92580, 92581, 92458, 92459, 92460, 92457, 92456, 92455, 92463, 92462, 92461, 92446, 92445, 92447, 92448, 92449, 92433, 92432, 92431, 92430, 92429, 92428, 92427, 92426, 92425, 92230, 92231, 92232, 92233, 92234, 92235, 92236, 92237, 92238, 92239, 92240, 92241, 92242, 92243, 92245, 92246, 92247, 92248, 92250, 92251, 92252, 92253, 92254, 92255, 92256, 92257, 92258, 92259, 92260, 92261, 92262, 92263, 92264, 92265, 92267, 92268, 92270, 92271, 92272, 92273, 92274, 92275, 92276, 92277, 92278, 92279, 92280, 92281, 92282, 92283, 92284, 92285, 92287, 92288, 92289, 92290, 92291, 92303, 92304, 92305, 92316, 92317, 92318, 92319, 92320, 92321, 92322, 92323, 92324, 92331, 92332, 92333, 92334, 92335, 92336, 92337, 92338, - 92339View allHide
ExoBrite™ CTB EV Staining Kits
30111-T, 30111, 30112-T, 30112, 30113-T, 30113, 30114-T, - 30114View allHide
RedDot™1 Far-Red Nuclear Stain, 200X in Water
40060-T, 40060, - 40060-1View allHide
NucSpot® Nuclear Stains
41040, 41040-T, 41033, 41033-T, 41036, 41036-T, 41037, 41037-T, 41034, 41034-T, 41035, 41035-T, 41038, - 41038-TView allHide

