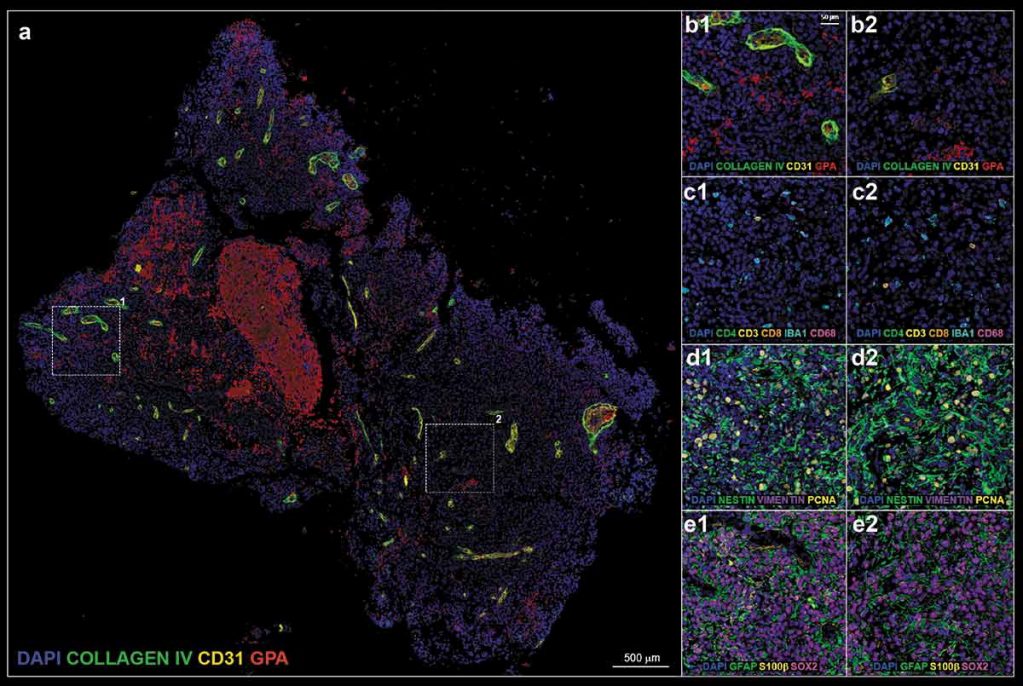
Glioblastoma (GBM) is a highly lethal form of brain cancer with significant morbidity and mortality rates. Treatment of GBM is particularly difficult in children due to higher susceptibility of neurotoxic side-effects from current treatments on developing brains. Recent advances in cancer immunotherapy, such as oncolytic herpes simplex virus (oHSV) therapy, may improve GBM survival rates by promoting anti-tumor immune responses that selectively kill tumor cells with limited side-effects. However, these methods are limited because of the expression of “checkpoint” proteins in human brain tumors that inhibit tumor infiltrating lymphocytes and prevent T cell activation. To maximize the efficacy of immunological methods, comprehensive information on the complex host-tumor relationship is needed. This includes assessment of both the functional and topographical states of infiltrating immune cell types in tumor biopsies. Currently, there are no methods that provide thorough phenotyping of GBM tumor/immunopathology in clinically relevant tissue.
In a recent issue of OncoImmunology, Bernstock et al. have developed a novel in situ multiplex immunofluorescence screening and imaging platform for clinical GBM samples before and after immunotherapy treatment. A tumor biopsy from an 11-year-old female with right parietal lobe glioblastoma was taken before and 3 months after oHSV treatment. The tumor tissue was then processed for immunostaining and treated with TrueBlack® Lipofuscin Autofluorescence Quencher to reduce intrinsic tissue autofluorescence. The sections were then sequentially stained with 4 antibody panels consisting of 18 validated biomarkers for GBM phenotyping. Results showed robust tumor infiltration of T-lymphocytes after oHSV treatment, suggesting the treatment amplified the anti-tumor immune response. However, expression of checkpoint proteins after treatment also increased significantly, suggesting co-treatment with checkpoint inhibitors may help maintain the anti-tumor response. Cumulatively, the authors demonstrate a viable multiplex biomarker screening platform useful for assessing human brain tumor response to immunologically centered therapies.
Learn more about Biotium’s TrueBlack® Lipofuscin Autofluorescence Quencher ideal for brain tissue background quenching as well as TrueBlack® IF Background Suppressor System for blocking non-specific secondary antibody binding.
Full Citation
Bernstock, J. D., Vicario, N., Rong, L., Valdes, P. A., Choi, B. D., Chen, J. A., … Friedman, G. K. (2019). A novel in situ multiplex immunofluorescence panel for the assessment of tumor immunopathology and response to virotherapy in pediatric glioblastoma reveals a role for checkpoint protein inhibition. OncoImmunology, 8(12), e1678921. https://doi.org/10.1080/2162402X.2019.1678921

