Alzheimer’s disease (AD) is a complex brain disorder. Age is the main driver but a combination of environmental, lifestyle and genetic factors influence its onset and pathology. The strongest genetic risk factor for AD is the E4 isoform of Apolipoprotein E (ApoE), a family of proteins involved in lipid and cholesterol transport. In the brain, APOE4 is associated with altered lipid and carbohydrate metabolism similar to that seen in AD patients. Recent studies also show increased accumulation of lipid droplets (LDs), specialized organelles containing triacylglycerols and cholesterol esters, in neurodegenerative conditions such as Parkinson’s and AD.
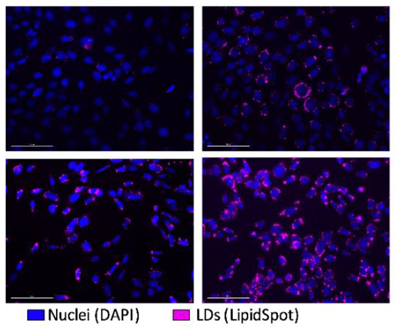
Farmer et al., in a recent publication in the journal Cells, characterize ApoE isoform-specific differences in LD formation and overall lipid metabolism in astrocytes (the main sites of ApoE production in the brain). Based on staining with the lipid droplet dye LipidSpot™ 610, astrocytes expressing E4 exhibit a significantly higher LD content but smaller individual LDs compared to those expressing E3 (normal isoform). This correlates with higher expression of the LD-associated protein perilipin-2 in E4 astrocytes. E4 astrocytes also show decreased exogenous fatty acid oxidation, and alterations in multiple measures of fatty acid metabolism. This study provides evidence linking the APOE genotype with astrocyte lipid storage and metabolism and can form the basis for developing therapies for AD and other neurodegenerative diseases.
Farmer BC, Kluemper J, Johnson LA. Apolipoprotein E4 Alters Astrocyte Fatty Acid Metabolism and Lipid Droplet Formation. Cells. 2019 Feb 20;8(2). pii: E182. doi: 10.3390/cells8020182.

