Studying the complex anatomy of the brain is a crucial task for understanding how the brain works. The intricate structures work together in brain functioning, and brain diseases can affect the morphology and the relationship of these structures. Because of the importance of studying the brain, as well as other whole organs, it is important to develop a standard protocol for observing these structures. Current techniques use resin embedding to preserve structural information of organs in three dimensions. However, the current method of resin embedding is unreliable due to incompatibilities with fluorescent dyes. The existing chemical treatment of resin embedding has led to quenching of fluorescence signal and poor signal-to-noise.
In a recent publication in Biomedical Optics, M. Ren et al. developed a protocol for improving the existing issues during resin embedding of large biological tissues. To reduce background fluorescence, they added TrueBlack® Lipofuscin Autofluorescence Quencher in the glycol methacrylate embedding medium. This method, named glycol methacrylate with TrueBlack® (GMA-T), was used to visualize structures labeled with fluorescent dyes and proteins. Whole brain and brain slices of C57 mice were sectioned using a vibratome either before or after, embedding using GMA-T.
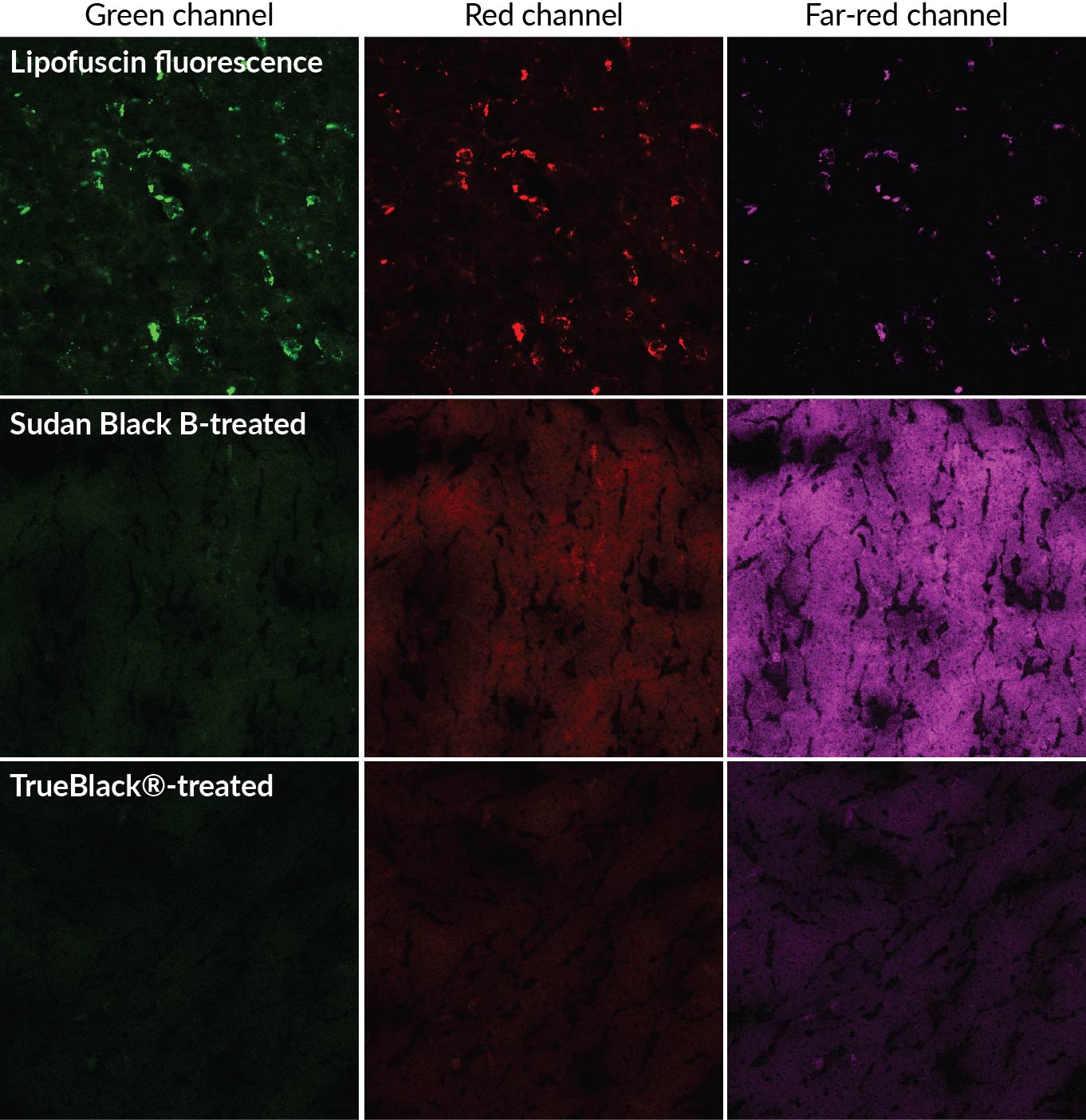
In comparison with the traditionally used autofluorescence quencher, Sudan Black B, TrueBlack® was more successful at inhibiting autofluorescence. The authors found that using 0.7% TrueBlack® in the GMA-T solution successfully retained over 80% of the intensity of the fluorescent dyes, as well as inhibited over 90% of the background fluorescence. The authors concluded that the use of Sudan Black B was the key factor contributing to poor results with fluorescent dyes in resin embedded samples. However, when Sudan Black B in the resin was replaced with TrueBlack®, fluorescent dyes could be imaged with sub-cellular resolution. The new approach to resin embedding of organs is important for imaging large-volume biological samples. Further implementation of this technique can be used to clear up current uncertainties about the anatomy of biological organs.
Learn more about Biotium’s unique TrueBlack® Lipofuscin Autofluorescence Quencher as well as our other TrueBlack® Background Reducers for immunofluorescence or western blotting.
Full Citation:
Ren, M., Tian, J., Sun, Q., Chen, S., Luo, T., Jia, X., Jiang, T., Luo, Q., Gong, H., & Li, X., (2021). Plastic embedding for precise imaging of large-scale biological tissues labeled with multiple fluorescent dyes and proteins. Biomed. Opt. Express 12, 6730-6745. https://doi.org/10.1364/BOE.435120

