Content #1
Content #1
Content #1
Fluorescence in situ hybridization (FISH) has offered an eloquent method for the detection of mRNA in whole-mount samples or sections for over 50 years. While low signal-to-background ratio was initially a limiting factor for FISH in certain fields, over the past two decades the incorporation of isothermal signal amplification mechanisms termed the hybridization chain reaction (HCR) has expanded FISH use into more diverse applications. After several iterations, the current third-generation in situ hybridization chain reaction (V3HCR) now allows clear detection of mRNA to subcellular or even single-molecule resolution. However, widespread lipofuscin autofluorescence, most often found in neuronal cell populations, interferes with HCR signal and can complicate analysis.
In a recent Current Protocols publication, May-Zhang et al. report a V3HCR protocol that allows the successful subcellular detection of specific mRNAs in mouse and human tissue cryosections. The protocol was optimized targeting mRNA in neuronal cells in adult intestinal tissue sections, but it can be applied to a range of tissues and sample thicknesses. After cryosectioning and fixing of tissues, the protocol is divided into three discrete day-long segments: 1) hybridization, 2) removal of unbound probe and signal amplification, and 3) washing, counterstaining, and mounting. To demonstrate its efficacy, the protocol was applied to sections of myenteric ganglia in adult mouse and human intestinal tissue to detect marker mRNAs for discrete categories of enteric neurons. After testing a panel of lipofuscin quenching methods, including quenching with cupric sulfate, bleaching in Dent’s fixative, detergent extraction, Murray’s clearing, treatment with 8% SDS, and standard Sudan Black blocking, Biotium’s TrueBlack® Lipofuscin Autofluorescence Quencher was determined to be the only acceptable option for overcoming lipofuscin autofluorescence without causing V3HCR signal loss. Along with demonstrating their detailed protocol in mammalian tissue sections, the authors also beautifully illustrate the difference in detection quality between samples treated with TrueBlack® and non-treated controls. This adds to extensive data showing the critical improvements achieved by using TrueBlack® when preparing samples prone to lipofuscin autofluorescence, such as human neuronal populations.
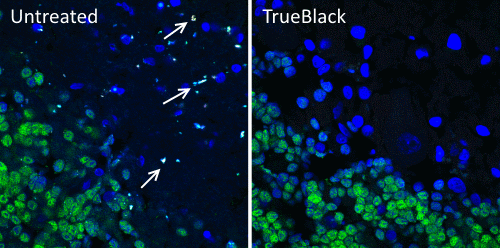
Check out Biotium’s full line of TrueBlack® Background Reducers for microscopy and western blotting as well as our extensive catalog of other immunofluorescence microscopy products including antifade mounting media, coverslip sealant, and more.
Full Citation
May‐Zhang, A. A., Benthal, J. T., & Southard‐Smith, E. M. (2022). Hybridization Chain Reaction for mRNA Localization in Single Cells from Mouse and Human Cryosections. Current Protocols, 2(5), e439.https://doi.org/10.1002/cpz1.439