Immunofluorescence staining offers a variety of advantages compared to colorimetric immunocytochemistry, most importantly, simultaneous multicolor detection of several targets. However, fluorescence detection in tissue samples can be hindered by strong background fluorescence. Autofluorescence can arise from exogenous and endogenous sources, including aldehyde fixation, extracellular matrix components, red blood cells, other pigmented cell types, and lipofuscin. Autofluorescence can make it all but impossible to distinguish specific signal from fluorescent antibodies and other probes from non-specific background.
Autofluorescence tends to be most severe in the shorter visible fluorescence wavelengths (blue, green, and red fluorescence). Tissue components exhibit less fluorescence in the far-red and near-infrared wavelengths. In some tissues, choosing fluorescent dyes with far-red and near-infrared emission can improve signal to noise (find out more about our far-red and near-infrared CF® Dyes). However, highly autofluorescent tissue components such as red blood cells and lipofuscin have broad emission spectra, and can also overwhelm far-red immunofluorescence signal.
A variety of methods for quenching autofluorescence have been reported, including amine quenching and borohydride treatment for aldehyde-fixed tissue (1), dye quenchers (1,2), or photobleaching of sections (3). While some researchers report success using various treatment combinations for particular tissues, for many tissues these methods are not effective enough for the detection of specific immunofluorescence staining. For example, while borohydride treatment is effective at reducing autofluorescence from glutaraldehyde fixation, it has been shown to increase red blood cell autofluorescence in formaldehyde-fixed tissue (1).
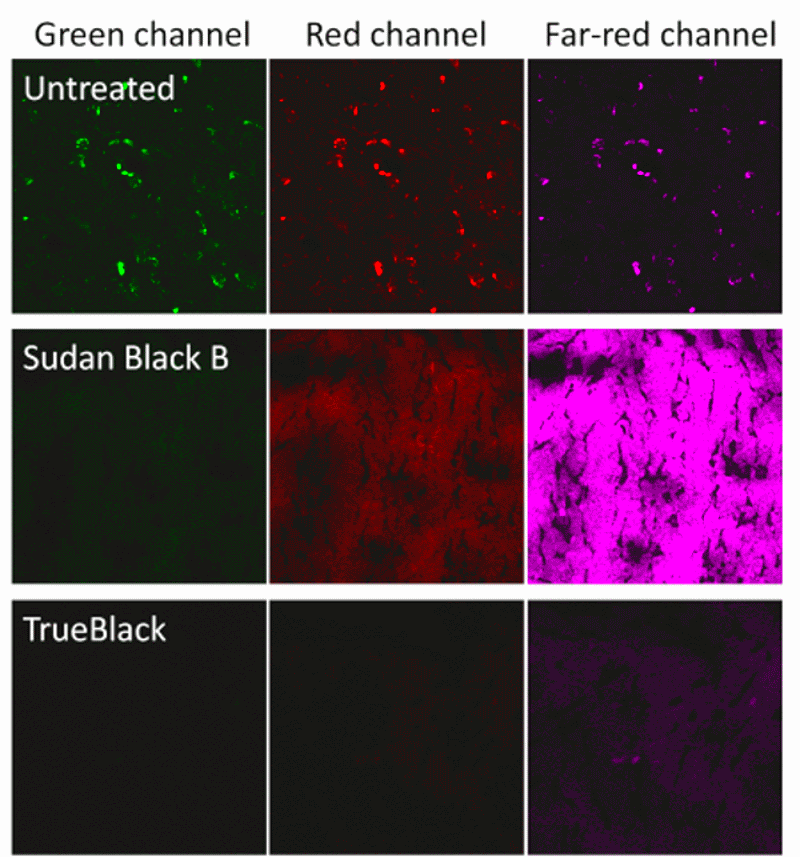
Fluorescence quenching by staining tissue with Sudan Black B is very effective at eliminating autofluorescence from lipofuscin (1, 2). Lipofuscin is a highly autofluorescent pigmented substance consisting oxidized lipids and proteins (4). Lipofuscin granules accumulate in lysosomes with age, and can present a major obstacle to fluorescence imaging of human tissues. Sudan Black B is highly lipophilic, and it binds to lipofuscin granules and quenches their fluorescence. While not a universal cure for tissue autofluorescence, it is indispensible for staining tissues with high accumulation of lipofuscin, like human brain. However, a major limitation of Sudan Black B is that it also fluoresces in the red and far-red wavelengths (Fig. 1 and Ref. 3), severely limiting the selection of dyes and filters available for multicolor detection.
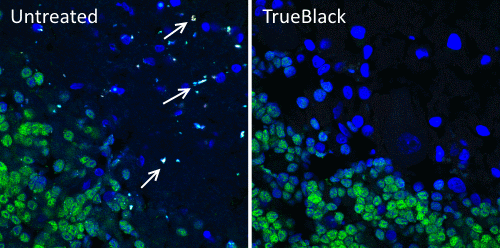
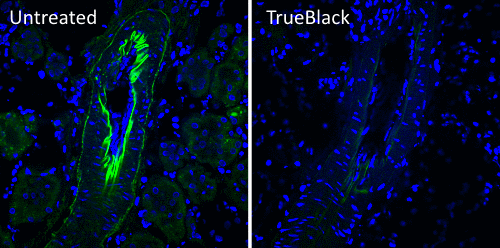
Biotium developed TrueBlack® Lipofuscin Autofluorescence Quencher (catalog no. 23007) as an improvement on Sudan Black B. Like Sudan Black B, TrueBlack® is a lipophilic black dye that quenches lipofuscin granules. However, TrueBlack® has much lower fluorescence in red and far-red wavelengths, making those channels available for multicolor imaging. TrueBlack® has been validated for lipofuscin quenching in human brain and retina (Fig. 2 and Refs. 5, 6). While TrueBlack® was designed specifically to quench lipofuscin, we’ve also found that it can substantially reduce other sources of background like autofluorescence from extracellular matrix and red blood cells in tissues like human and rodent kidney, as previously reported for Sudan Black B (Fig. 3 and Ref. 7).
Protocols for quenching autofluorescence with TrueBlack®
TrueBlack® can be applied before antibody staining to minimize effects on antibody binding and fluorophore fluorescence. TrueBlack® preferentially quenches lipofuscin, with slight quenching of fluorescent dyes, so if necessary, it also can be applied after immunofluorescence staining is complete. The pre-treatment and post-treatment protocols for TrueBlack® are outlined below.
Pre-treatment protocol (preferred):
Important: all steps after TrueBlack® treatment must be carried out without detergent. If detergent is required in your antibody incubation or wash steps, use the post-treatment protocol.
- Pre-process tissue sections for staining. For FFPE sections, deparaffinize and perform antigen retrieval steps if necessary. For cryosections, air dry slides and fix in the fixative recommended for your antigen(s).
- Permeabilize sections with detergent, if required.
- Rinse slides in PBS.
- Prepare TrueBlack® treatment solution. TrueBlack® is supplied as a 20X stock solution. Dilute the stock 1:20 in 70% ethanol to make 1X TrueBlack® treatment solution.
- Blot excess buffer away from sections and place slides on a level staining rack. Cover each section completely with a drop (50-100 uL) of 1X TrueBlack® solution. Incubate for 30 seconds.
- Rinse slides 3X with PBS
- Perform blocking, antibody incubations, and washes according to your standard immunofluorescence protocol. Be sure to use buffers that don’t contain detergent. If detergent is required during these steps, use the TrueBlack® post-treatment protocol.
- Mount slides in aqueous antifade mounting medium with or without DAPI.
Post-treatment protocol
Note: Post-treatment may result in lower immunofluorescence signal, but it is compatible with protocols that require blocking or washing with detergent.
- Pre-process tissue sections and perform immunofluorescence staining according to your usual protocol. Counterstaining of nuclei can be performed before or after the TrueBlack® treatment.
- Prepare TrueBlack® treatment solution. TrueBlack® is supplied as a 20X stock solution. Dilute the stock 1:20 in 70% ethanol to make 1X TrueBlack® treatment solution.
- After the final wash step of your staining protocol, blot excess buffer away from sections and place slides on a level staining rack. Cover each section completely with a drop (50-100 uL) of 1X TrueBlack® solution. Incubate for 30 seconds.
- Rinse slides 3X with PBS
- Mount slides in aqueous antifade mounting medium with or without DAPI.
References
- Baschong, W., Suetterlin, R. and Laeng, R.H. (2001). Control of autofluorescence of archival formaldehyde-fixed, paraffin-embedded tissue in confocal laser scanning microscopy (CLSM). J Histochem Cytochem. 49(12): 1565.
- Romijn, H.J., van Uum, J.F.M., Breedijk, I., Emmering, J., Radu, I., and Pool, C.W. (1999). Double immunolabeling of neuropeptides in the human hypothalamus as analyzed by confocal laser scanning fluorescence microscopy. J Histochem Cytochem 47(2): 229.
- Neumann, M. and Gabel, D. (2002). Simple Method for Reduction of Autofluorescence in Fluorescence Microscopy. J Histochem Cytochem. 50(3): 437.
- Hohn, A. and Grune, T. (2013). Lipofuscin: formation, effects and role of macroautophagy. Redox Biol 1(1): 140.
- Fosso, M.Y., McCarty, K., Head E., Garneau-Tsodikova, S., and LeVine, H. 3rd. (2015). Differential Effects of Structural Modifications on the Competition of Chalcones for the PIB Amyloid Imaging Ligand-Binding Site in Alzheimer’s Disease Brain and Synthetic Aβ Fibrils. ACS Chem Neurosci 7(2): 171.
- Chan, T., Zhu, L., Madigan, M.C., Wang, K., Shen, W., Gillies, M.C., Zhou, F. (2015). Human organic anion transporting polypeptide 1A2 (OATP1A2) mediates cellular uptake of all-trans-retinol in human retinal pigmented epithelial cells. Br J Pharmacol 172(9): 2343.
- Sun, Y., Yu, H., Zheng, D., Cao, Q., Wang, Y., Harris, D., Wang, Y. (2011). Sudan Black B Reduces Autofluorescence in Murine Renal Tissue. Arch Pathol Lab Med 135: 1335.
