Content #1
Content #1
Content #1

Fei Mao, PhD. CEO, CSO and Co-Founder of Biotium
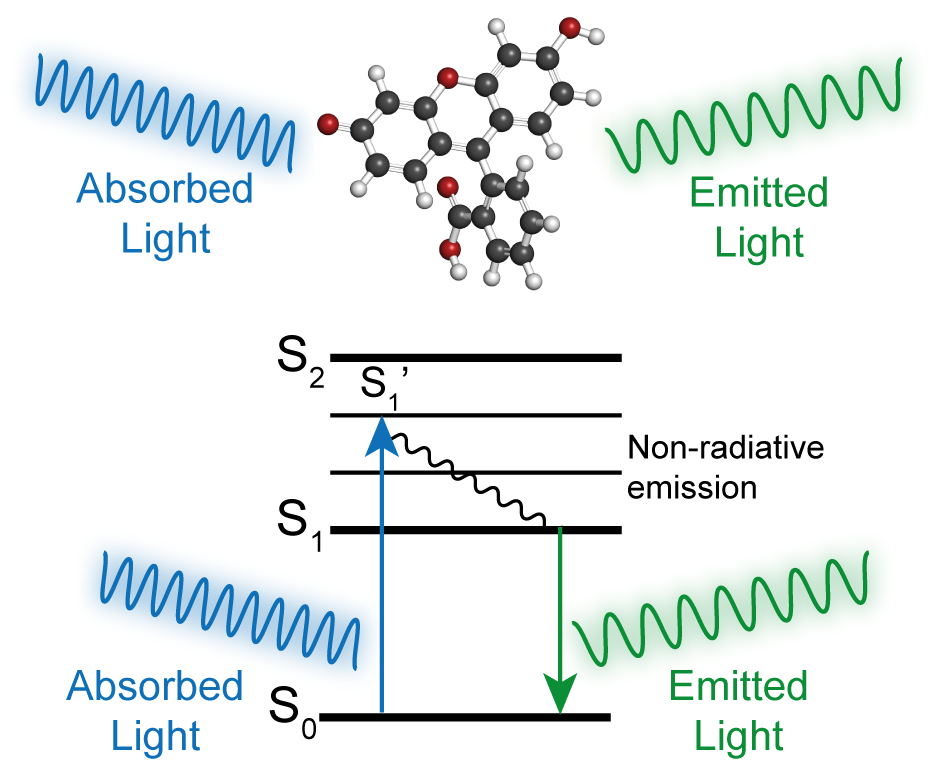
Figure 1. Jablonski Diagram of excitation and emission of a fluorescent dye.
A molecule’s brightness is determined by two metrics, molar absorptivity (ε) and quantum yield (Ф). The molar absorptivity, also known as a molar extinction coefficient, is a measure of how much light a molecule absorbs at a given wavelength. Meanwhile, the quantum yield is a ratio between how much light is emitted versus absorbed. The brightness of a molecule can be determined by multiplying the fluorescence quantum yield by the molar absorptivity.
Brightness = Quantum Yield (Ф) X Molar Absorptivity (ε)
Despite the simplicity of this equation, the process is a bit more complex. A molecule’s fluorescence profile and quantum yield can depend heavily on the environment. For example, dye-protein conjugations can lead to nonfluorescent aggregates known as H-aggregates that drastically reduce brightness. This effect increases as more dye molecules are conjugated per protein. Other environmental factors that can influence a molecule’s fluorescence profile are the type of solvent used, dye concentration, and pH of the medium.
Brightness is just one piece of a much larger puzzle. In addition to fluorescence intensity, chemists must consider photostability, chemical stability, sources of background fluorescence, and other factors that determine how the dye will perform within a biological context. This poses a serious challenge for chemists, where chemical modifications intended to improve one characteristic may be detrimental to others. For example, in our last post we left off with the issue of sulfonation, a chemical modification commonly found in today’s Cy® and Alexa Fluor® dyes. Sulfonation supplies negative charges to the molecule which improves solubility and reduces dye aggregation. However, the negative charges also increase non-specific binding and background fluorescence. Essentially, we’ve traded one desirable attribute for another less desirable one. So, where do we go from here?
Scientists at Biotium have pushed the envelope of modern fluorescence by developing CF® Dyes, a new family of dyes with superior brightness, photostability and signal-to-noise. But what makes the CF® Dyes so unique? Let’s focus on two novel chemical modifications that set CF® Dyes apart from the competition: PEGylation and the rhodamine-imidazole substituent.
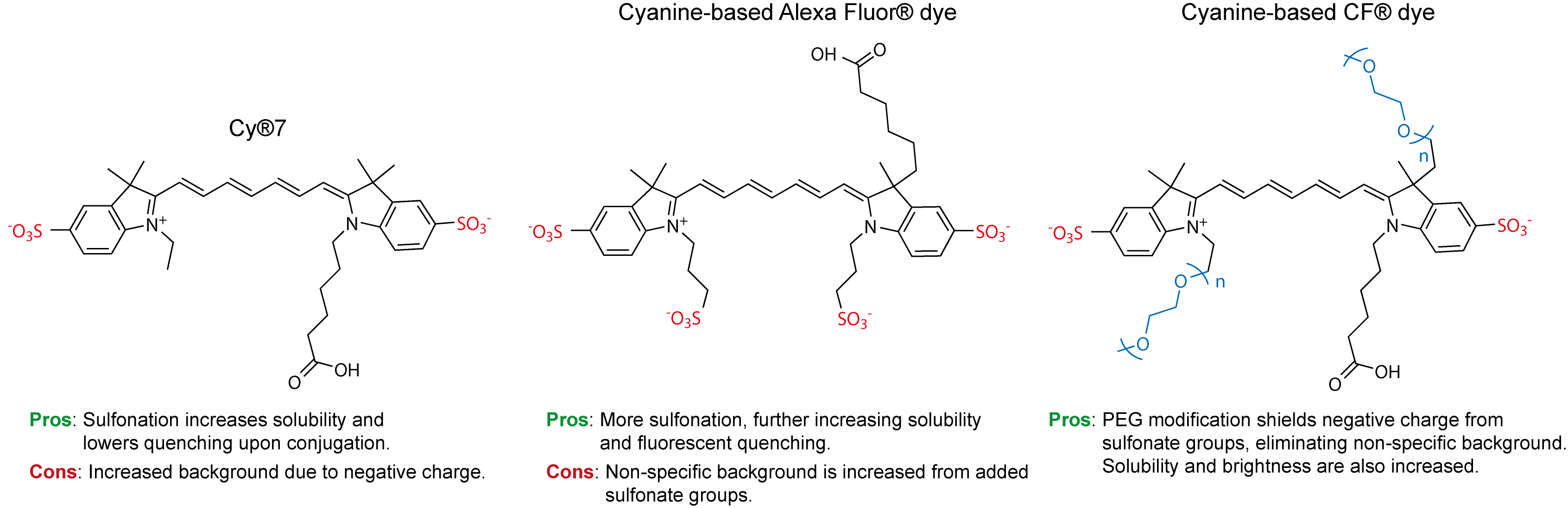
Figure 2. Evolution of cyanine dyes.
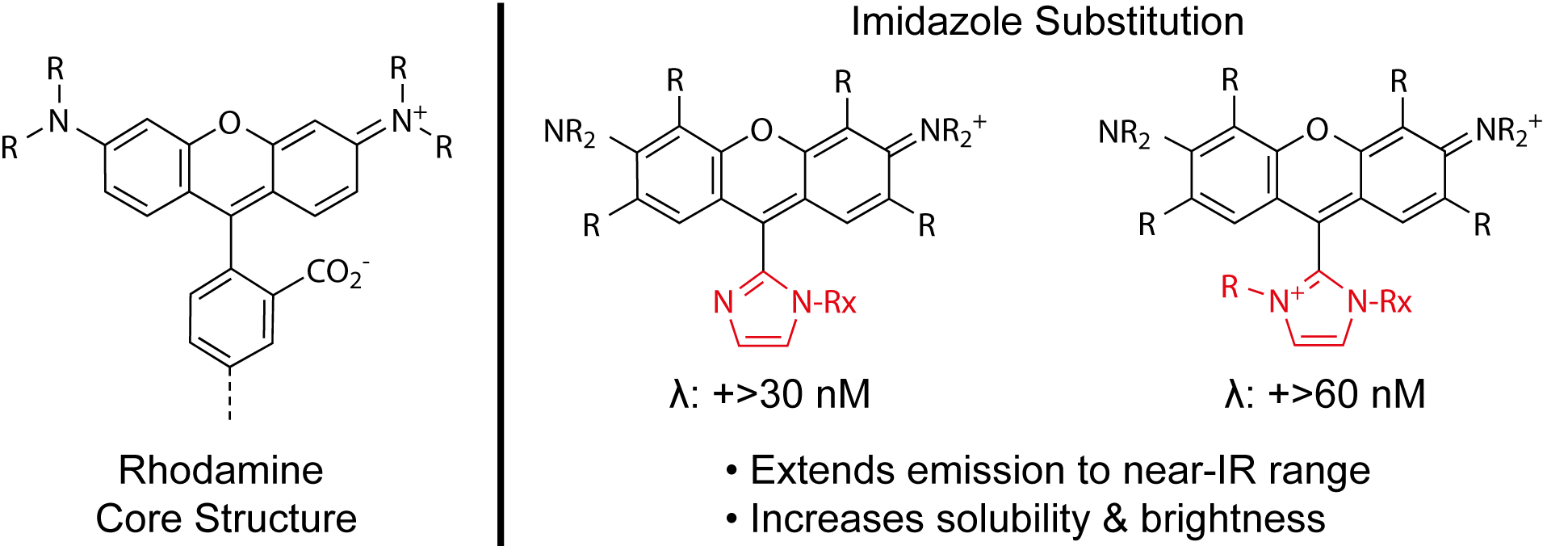
Figure 3. Imidazole substitution of rhodamine-based dyes.
After this in-depth look at the chemistry behind our CF® Dyes, you might be wondering what does “CF” stand for? CF® was initially an abbreviation for “Cyanine-based Fluorescent dyes”. These were the first patented CF® Dyes based on cyanine dye structures. 10 years and more than two dozen dyes later, the CF® dye portfolio encompasses multiple dye core structures spanning the fluorescence spectrum from UV to near-IR. Today, we believe “CF” more aptly stands for Clear Fluor: dyes that produce superior signal-to-noise.
At the end of the day, our products are used to produce images representative of complex biological systems. Proper interpretation of those images requires clarity and trust in the signal the dye produces. With the chemical advancements described here we believe our dyes earn that trust. Today, our fluorescent CF® Dyes lead the industry, offering superior brightness, photostability, and biocompatibility from blue to near-IR.
Learn more about Biotium’s CF® Dyes and our other innovative fluorescent technologies.
Eric Torres holds a PhD in Biochemistry and Molecular Biology from the University of California, Los Angeles. His dissertation research focused primarily on mechanisms of mitochondrial protein import. He is the current Marketing Manager at Biotium.