Fluorescence microscopy is becoming increasingly important in research and clinical diagnostics with the development of novel dyes and imaging techniques. However, autofluorescence remains an underlying issue, especially in studies that rely on fixed mammalian tissues. Appearing as strong background fluorescence, across multiple channels, it interferes with the signal of several commonly used fluorescent reporters and complicates data analysis. Extracellular matrix components, red blood cells (RBCs), lipofuscin, pigmented cells and fixation methods all can contribute to autofluorescence. Several quenchers and tissue treatment methods have been developed as countermeasures, but with limited success. Tissue or whole organism perfusion to flush-out interfering RBCs have limited use for embryonic tissues or clinical samples.
In a recent report in the Current Protocols in Neuroscience, the authors demonstrate how TrueBlack® efficiently quenches RBC autofluorescence across multiple wavelengths, in fixed embryonic tissue, without interfering with immunofluorescent signal intensity or introducing background staining. They also demonstrate that TrueBlack® is compatible with multicolor immunofluorescence staining as opposed to commonly used quenchers such as Sudan Black, and provide detailed information on optimal concentrations, incubation times and multiple usage conditions for routine immunofluorescence microscopy.
To read the original article, click here.
Whittington, N. C., & Wray, S. (2017). Suppression of red blood cell autofluorescence for immunocytochemistry on fixed embryonic mouse tissue. Current Protocols in Neuroscience, 81,2.28.1–2.28.12. doi: 10.1002/cpns.35
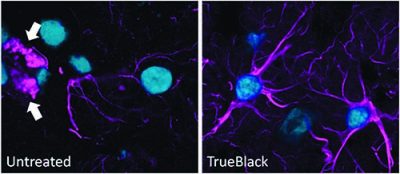
Quenching of lipofuscin autofluorescence using TrueBlack® post-treatment after immunofluorescence staining of methanol-fixed cryosections of human cerebral cortex stained with rabbit anti-GFAP followed by CF®640R goat anti-rabbit and DAPI mounted in EverBrite™ mounting medium.
Learn more about Biotium’s TrueBlack® lipofuscin autofluorescence quencher. Also see our Tech Tip: Battling Tissue Autofluorescence.

