Targeted drug delivery to pathological sites could reduce systemic toxicity, clinical side effects, and the high dosage requirement of standard treatment protocols. Unfortunately, the lack of methods for efficient targeted delivery remains a bottleneck in drug administration. The optimal targeting strategy is delivery into the therapeutic molecule’s specific intracellular active site or organelle. Mitochondria are key therapeutic targets for intracellular drug delivery due to their broad role in cell function as well as certain neurological diseases and cancers. Nevertheless, hydrophobicity, dense double membranes, and high negative membrane potential make mitochondria a difficult target. For over a decade now, much effort has been made to further develop mitochondria-targeted delivery systems.
Liu et al. report a new development towards overcoming hurdles associated with mitochondria-specific drug delivery in a topical ACS Nano publication. Specifically, they synthesized and compared the targeted delivery potential of shell-fluorinated and core-fluorinated micelles from two fluoroamphiphile analogs, phosphatidylethanolamine-polyethylene glycol-fluorocarbon and polyethylene glycol-fluorocarbon. Using fluorescent dye or the model drug vitamin E succinate (VES) as cargo, they tested the intracellular targeting capabilities of fluorinated micelles in human cell lines, cellular spheroids, and in murine cancer models. Biotium’s potential-independent MitoView™ Green and potential-dependent MitoView™ 633 fluorescent mitochondrial dyes (Figure 1) were used in key experiments to visualize colocalization of fluorescent cargo molecules with mitochondria, while the JC-1 assay and induced depolarization allowed the team to determine mitochondrial membrane potential and the effects of depolarization on cargo localization. Their results suggested that core-fluorinated micelles experienced clathrin-mediated endocytosis followed by rapid endosomal escape and showed 20X improved cellular uptake of the cargo compared to their shell-fluorinated counterparts. The authors hypothesized that micellar polyethylene glycol-fluorocarbon interacted efficiently with mitochondrial hydrophobic structures, resulting in a high potential-independent mitochondrial affinity. For an in vivo follow-up study, the team used an immunocompetent murine model of breast cancer to test for targeted delivery. After intravenous injection, micellar biodistribution was evaluated and showed significantly higher in vivo retention than free VES. Additionally, after 2 weeks, fluorinated micellar VES injections inhibited tumor size by ~75% and tumor weight by ~65% compared to the saline control group. Together these results present charge-neutral fluoroamphiphile micelles as a promising tool for mitochondrial targeting.
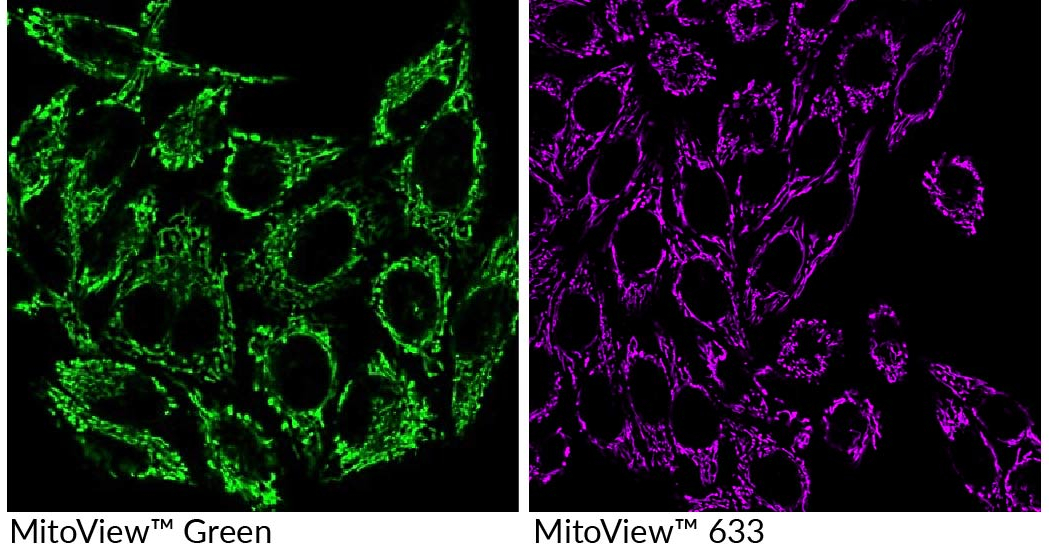
Along with our MitoView™ products, Biotium carries a range of organellar and nuclear stains, abundant primary antibody options, and other reagents for immunofluorescence microscopy.
Full Citation
Liu, Y., Zhang, J., Tu, Y., & Zhu, L. Potential-independent intracellular drug delivery and mitochondrial targeting. ACS nano, 16(1), 1409-1420 (2021). https://doi.org/10.1021/acsnano.1c09456

