Cardiovascular disease remains the most common cause of death and disability worldwide. It includes a range of conditions involving various heart muscle or blood vessel problems that restrict oxygen and blood flow to organ systems. Limited blood flow to the heart muscle results in myocardial ischemia (I), and rapid restoration of blood and oxygen supply by reperfusion (R) is the best remedy to limit the damage. Paradoxically, however, while a significant proportion of cardiac myocytes survive the ischemic episode, they die due to reperfusion injury.
In a recent PLoS ONE study, Sciuto et al., use multiple fluorescent reporters to establish the chronology of pathophysiological changes associated with I/R damage in neonatal rat ventricular myocytes. MitoView™ 633 was used to monitor changes in inner mitochondrial membrane potential in combination with other indicators for cytosolic calcium and zinc, as well as for sarcolemmal membrane integrity. A genetically expressed mitochondrial calcium indicator was also validated by MitoView™ 633 colocalization and used to monitor calcium accumulation in the organelle. This study speculates that a non-selective putative pore/channel in the sarcolemma is responsible for abnormal calcium influx leading to cell death, which may provide a basis for developing effective intervention strategies to improve outcomes of myocardial I/R.
Learn more about our MitoView™ dyes and other mitochondrial stains. We also offer a full range of cellular stains for live and fixed cell imaging.
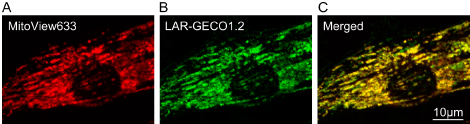
Credit: 2019 Sciuto et al. https://doi.org/10.1371/journal.pone.0212076.g001
Sciuto KJ, Deng SW, Moreno A, Zaitsev AV (2019) Chronology of critical events in neonatal rat ventricular myocytes occurring during reperfusion after simulated ischemia. PLoS ONE 14(2): e0212076. https://doi.org/10.1371/journal.pone.0212076.

