Content #1
Content #1
Content #1
The most common use of GelRed® and GelGreen® nucleic acid stains is in pre-cast agarose gels, where the dyes are added to molten agarose during gel preparation. However, due to their larger size designed to improve safety, band migration of DNA in pre-cast gels may be affected. Some samples, such as restriction digested DNA can migrate abnormally in GelRed® or GelGreen® precast gels.
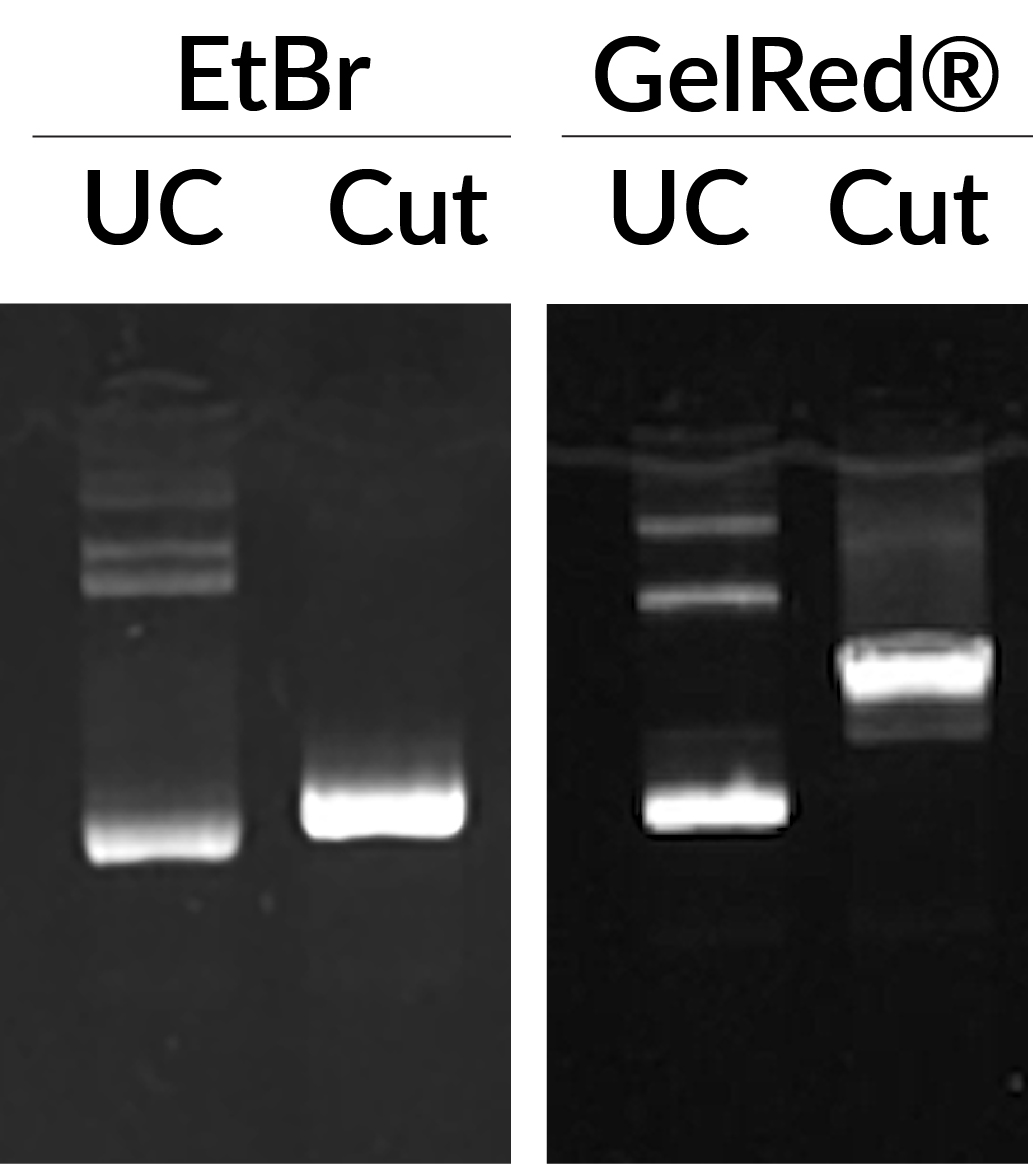
Ethidium bromide (EtBr) and GelRed® in precast staining of uncut pGL3 plasmid (UC), or pGL3 plasmid cut with Mul1 (Cut). GelRed® displays abnormal band migration of cut pGL3 plasmid compared to EtBr. Each lane was loaded with 75 ng DNA.
This is why overloading DNA results in smearing, smiling, or curvy bands, and aberrant DNA migration. The recommended loading amount for samples of known concentration or commercial ladders/DNA markers is 50-200 ng/lane. For samples of unknown concentration, loading 1/2 - 1/4 the usual amount of DNA usually solves migration issues.
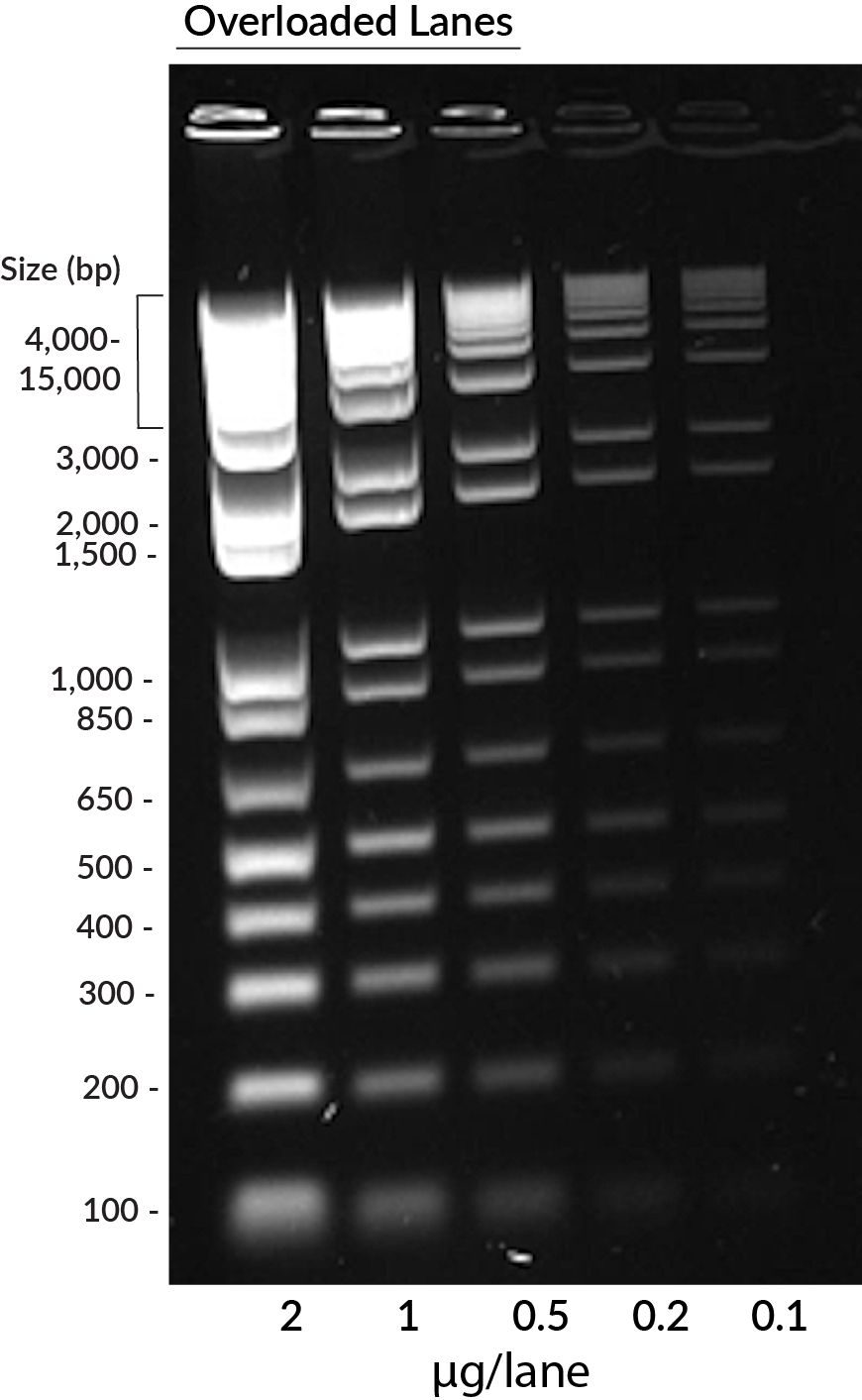
GelRed® in precast staining of 1 kb DNA ladder (Invitrogen) on a 1% agarose TBE gel. Ladder was loaded in the amounts of 2 ug, 1 ug, 0.5 ug, 0.2 ug, and 0.1 ug per lane from left to right. Overloaded lanes with more than 0.2 ug DNA displayed smiling and smearing bands.
Low DNA amounts (0.1 ng or lower) can be reliably detected. Loading high DNA amounts is not required, and can result in issues with DNA migration. Loading lower DNA amounts gives better separation and sharper bands, saving on precious samples, DNA ladder and conserving costs.
Loss of staining, or faint staining of low molecular weight DNA bands in not an issue as GelRed® and GelGreen® do not migrate as fast as ethidium bromide through the gel. It is not necessary to add additional dye to the running buffer.
As GelRed® and GelGreen® are larger dyes, high molecular weight DNA separates better on lower percentage agarose gels.
Tris-borate-EDTA (TBE) buffer has a higher buffering capacity than Tris-acetate-EDTA (TAE) buffer. For this reason we recommend using TBE buffer if you are using TAE buffer to improve band resolution.
Precast gels containing GelRed® or GelGreen® can be stored for future use for up to a week, or month respectively. We recommend storing gels at room temperature, protected from light.
Weak fluorescence, decreased dye performance over time, or a film of dye remaining on the gel after post-staining are indications that the dyes may have precipitated out of solution. Warm the dye solutions to ~50°C, and mix well by vortexing to re-dissolve precipitates.
Because high affinity nucleic acid binding dyes like GelRed® and GelGreen® can affect DNA migration during electrophoresis, post-staining of gels is highly recommended. Post-staining is also recommended if (a) loading higher than the recommended amount of DNA is needed, (b) you are using loading buffers containing SDS, which can contribute to band smearing, or (c) if band migration issues in precast gels with GelRed® or GelGreen® are persistent. Post-staining can take 5-30 min, depending on how much DNA is present, and the solution can be reused.