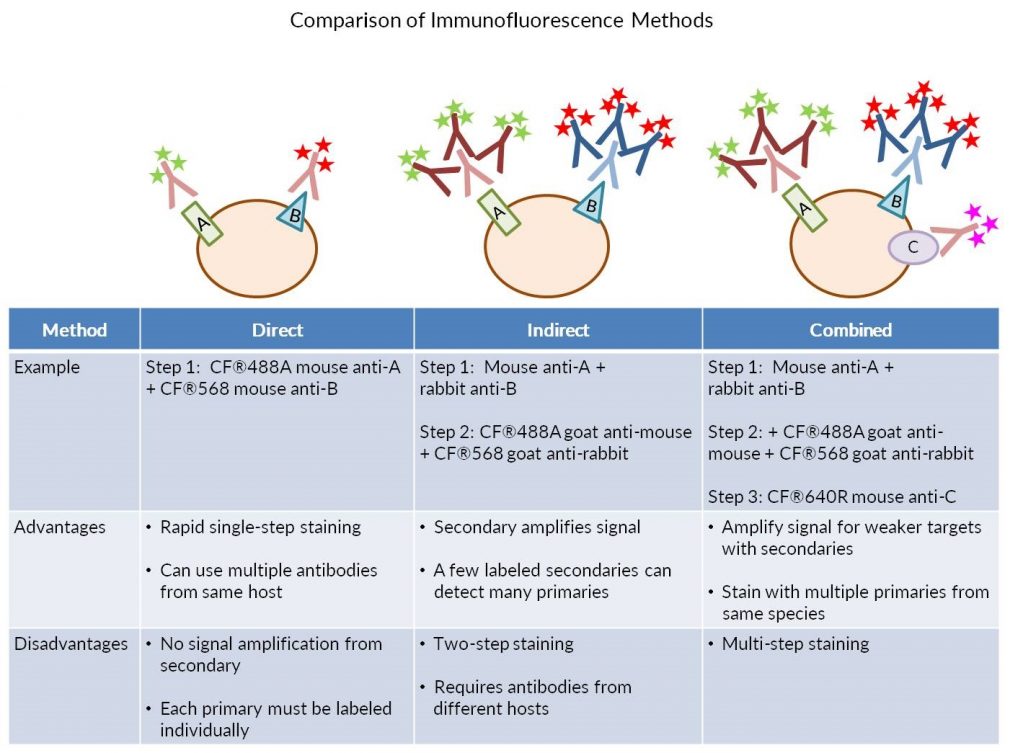
Immunofluorescence staining (IF) can be performed in two ways, by direct or indirect detection. Direct IF uses a dye-conjugated antibody to stain the target protein. Indirect IF involves first binding the primary antibody to the target, then detecting the primary antibody using a conjugated secondary antibody. Indirect IF offers the advantage of higher sensitivity. Each conjugated antibody molecule usually bears 4-6 fluorescent dyes. With direct IF using monoclonal antibodies, only one antibody may bind to its target antigen on the protein, tagging it with 4-6 dye molecules.
With indirect IF, multiple secondary antibodies bind to each primary antibody, greatly increasing the number of dyes associated with the target protein. However, direct IF offers the advantage of being able to stain a sample with multiple primary antibodies from the same host species simultaneously.
Secondary antibodies will still recognize a dye conjugated primary antibody, therefore a directly labeled antibody cannot be combined with an unconjugated primary from the same species in a single staining step, because the labeled secondary antibody will detect both the labeled and unlabeled primary antibodies. However, staining can be performed sequentially to allow indirect detection of the first primary antibody using a secondary, followed by staining with a directly labeled primary antibody from the same species, without cross-reactivity with the secondary. The following protocol provides an example of sequential staining with two unconjugated primary antibodies from rabbit and mouse hosts, followed by one directly labeled mouse primary. It can be adapted for other combinations of primary antibody host species.
Protocol for combined direct and indirect immunofluorescence
1. Perform fixation, permeabilization, and blocking of your samples according to the preferred protocol for your primary antibodies.
Note: See our Protocols for Antibody-Based Detection for more information on fixation, permeabilization, and blocking.
2. Incubate samples with unlabeled mouse and rabbit primary antibodies diluted in blocking buffer at the manufacturer’s recommended concentration for 2 hours at room temperature or 4°C overnight.
3. Rinse samples three times with PBS, and then perform 3 x 5 minute washes with PBS.
4. Incubate samples with fluorescent anti-mouse and anti-rabbit secondary antibodies diluted in blocking buffer for 30 minutes to 2 hours at room temperature, protected from light. Typically, fluorescent secondary conjugates are used at a final concentration of 1-2 ug/mL.
Note: Be sure to use highly cross-adsorbed secondary antibodies with minimal cross-reactivity for all antibody host species. If staining human or animal tissue sections that may contain endogenous immunoglobulins, make sure the secondary antibodies also have minimal cross-reactivity to the tissue itself.
5. Rinse samples three times with PBS, then perform 3 x 5 minute washes with PBS, protected from light.
6. Incubate samples with directly labeled mouse primary antibody diluted in blocking buffer for 2 hours at room temperature. Nuclear counterstains or labeled phalloidin may be included at this step.
Note: You may need to use a higher concentration of directly labeled primary antibody than you would use for indirect immunofluorescence.
7. Rinse samples three times with PBS, then perform 3 x 5 minute washes with PBS, protected from light.
8. Mount samples in antifade mounting medium and image.
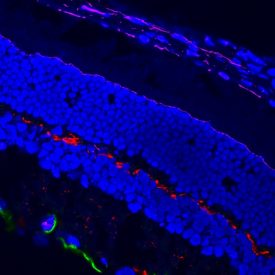
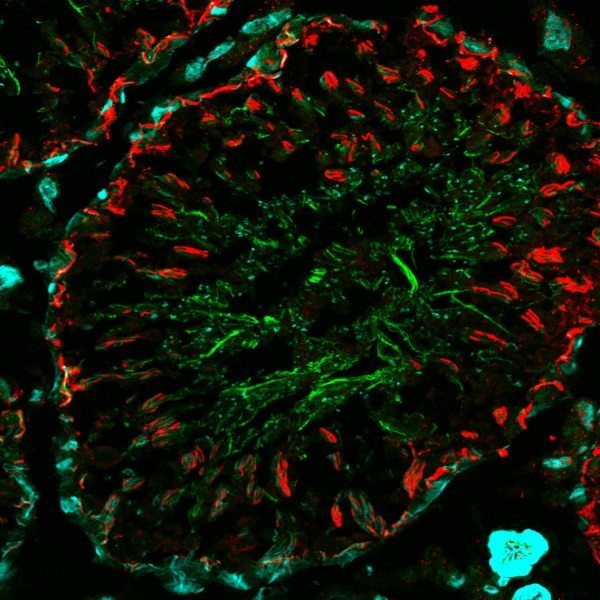
Example data
Related Products
Biotium offers a wide selection of highly cross-adsorbed secondary antibodies conjugated to our bright and photostable CF® dyes. We also offer more than 1000 primary antibodies, available in purified format or with your choice of 13 CF® dye colors. In addition, our Mix-n-Stain™ antibody kits provide a fast and simple way for you to conjugate your own primary antibody with a wide selection of CF® dyes. Also, be sure to see our accessory products for immunofluorescence, including TrueBlack® Lipofuscin Autofluorescence Quencher, EverBrite™ antifade mounting media, and CoverGrip™ Coverslip Sealant.