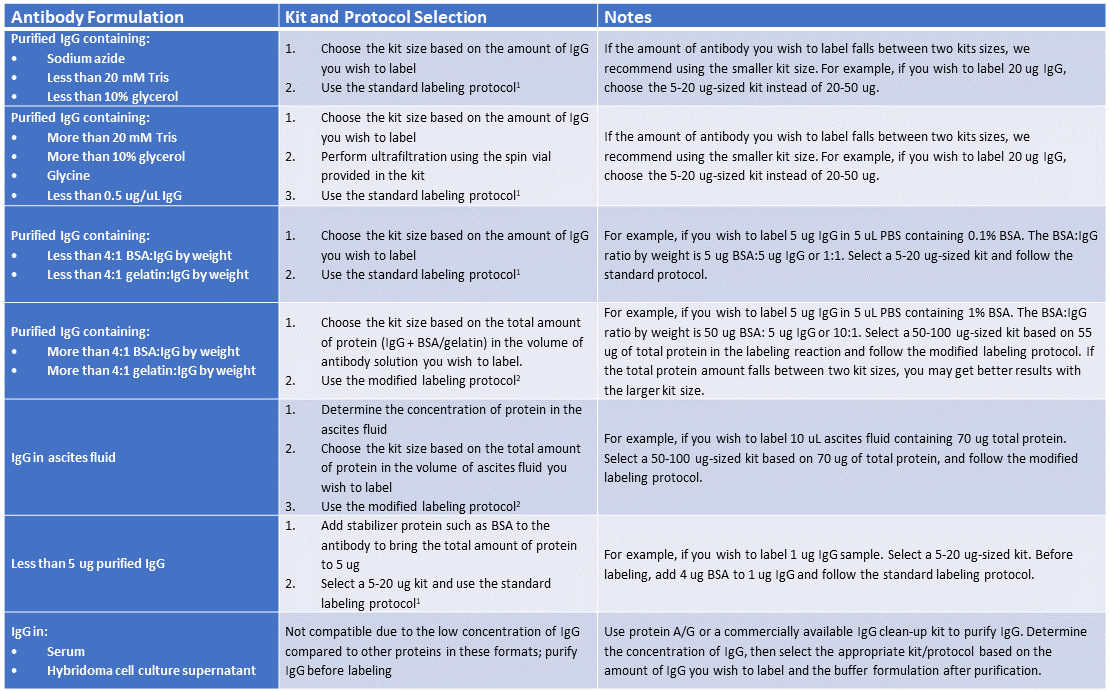
Content #1
Content #1
Content #1
If you're struggling to find suitable antibodies for your experiment, directly labeling your own antibodies can be a great option that offers more flexibility. Direct labeling allows you to choose any combination of antibody and label, including combinations that are not commercially available. Direct labeling also allows you to only label the amount of antibody you need, saving unlabeled antibody for other labels or applications. In addition, using directly conjugated primary antibodies enables the use of multiple primary antibodies from the same host and excludes the need for an additional incubation step required for secondary antibodies (see Tech Tip: Combined Direct and Indirect IF Using Primary Antibodies from the Same Host).

Mix-n-Stain™ Antibody Labeling Kits are designed to dramatically simplify the process of antibody labeling, allowing researchers to quickly conduct their experiment without wasting time optimizing antibody conjugations. A key feature of Mix-n-Stain™ labeling kits is the patented buffer system that tolerates stabilizer proteins and other common antibody buffer additives that interfere with conventional conjugation reactions unless the antibody is purified beforehand. In this article, we present technical tips and data for using Mix-n-Stain™ CF® Dye Antibody Labeling Kits with antibody solutions containing common buffer additives, including stabilizer proteins like BSA. For detailed protocols and for information about other Mix-n-Stain™ labeling kits (i.e., for fluorescent proteins, tandem dyes, or other labels), please refer to the specific product information sheet.
Steps before selecting your kit and protocol
Mix-n-Stain™ Compatibility and Kit Selection Guidelines
Labeling small IgG amounts (~1 ug) with Mix-n-Stain™
It is important to know the composition of your antibody solution when selecting a Mix-n-Stain™ kit size and protocol for labeling. Mix-n-Stain™ CF® Dye Antibody Labeling Kits can be used with antibody solutions of purified 0.5-1 mg/mL IgG containing sodium azide, trehalose, sucrose, a ratio of less than 4:1 BSA or gelatin to IgG by ug amount, and up to 20 mM Tris and 10% glycerol with no modifications to the labeling protocol.
For antibody solutions with additives within the tolerated limits, Biotium recommends the standard Mix-n-Stain™ labeling protocol (Protocol B on the Mix-n-Stain™ Antibody Labeling Kits Product Information Sheet). If buffer additives fall outside the recommended concentrations, purification, ultrafiltration, or other modifications to the labeling protocol may be required. In these instances, Biotium recommends the modified protocol (Protocol C on the Mix-n-Stain™ Antibody Labeling Kits Product Information Sheet), which contains an optional ultrafiltration step that is performed just before adding the storage buffer to the labeled conjugate.
Note: If you do not know the concentration of BSA, gelatin, or ascites fluid in your antibody solution, you can obtain the total protein concentration by measuring the OD280 and calculate the ratio of total protein to IgG.
Mix-n-Stain™ CF® Dye Antibody Labeling Kits have been proven to tolerate excess BSA, gelatin, or ascites fluid in the conjugation reaction without sacrificing performance of the labeled antibody. In addition, because CF® Dyes are highly water soluble, the presence of stabilizer proteins in the labeling reaction has minimal effect on background fluorescence because the proteins are readily washed away during immunofluorescence staining.
Labeling with Mix-n-Stain™ CF® Dye Antibody Labeling Kits in the presence of 10-fold excess BSA or gelatin using the modified protocol demonstrated no reduction in signal-to-noise during flow cytometry analysis of labeled anti-transferrin antibody in Jurkat cells (Figure 1). Similar results were seen when visualizing Mix-n-Stain™ labeled anti-transferrin antibody labeled in the presence of 10-fold excess BSA and gelatin in immunofluorescence microscopy in HeLa cells (Figure 2). Comparable performance was also shown in HeLa cells between indirect detection of anti-tubulin antibody using a secondary antibody and direct detection using CF®488 anti-tubulin that was labeled with Mix-n-Stain™ in the presence of excess BSA or in unpurified ascites fluid containing approximately 5 ug/mL IgG and 15 ug/mL other protein (Figure 3).
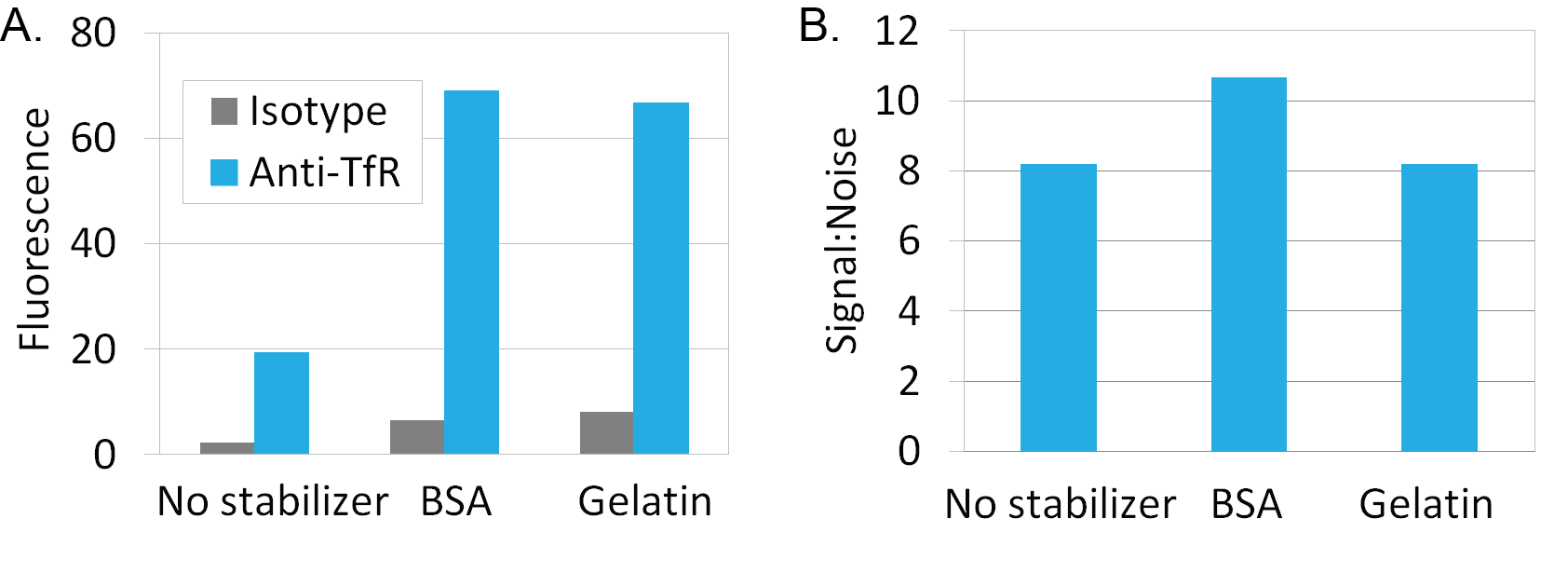
Figure 1. Mix-n-Stain™ labeling in the presence of 10-fold excess BSA or gelatin to IgG by weight using the modified Mix-n-Stain™ protocol. Mouse anti-transferrin receptor IgG or mouse IgG isotype control from BD Pharmingen were labeled using CF647 Mix-n-Stain™ kits. Intracellular staining of Jurkat cells was analyzed in channel FL3 of a BD FACSCalibur flow cytometer. Antibodies labeled with the modified protocol in the presence of stabilizer showed comparable signal to noise ratio compared to conjugates labeled without stabilizer using the standard labeling protocol.
A. Graph shows the geometric mean fluorescence of the cell populations.
B. Graph shows signal to noise ratio (anti-TfR fluorescence/isotype fluorescence).
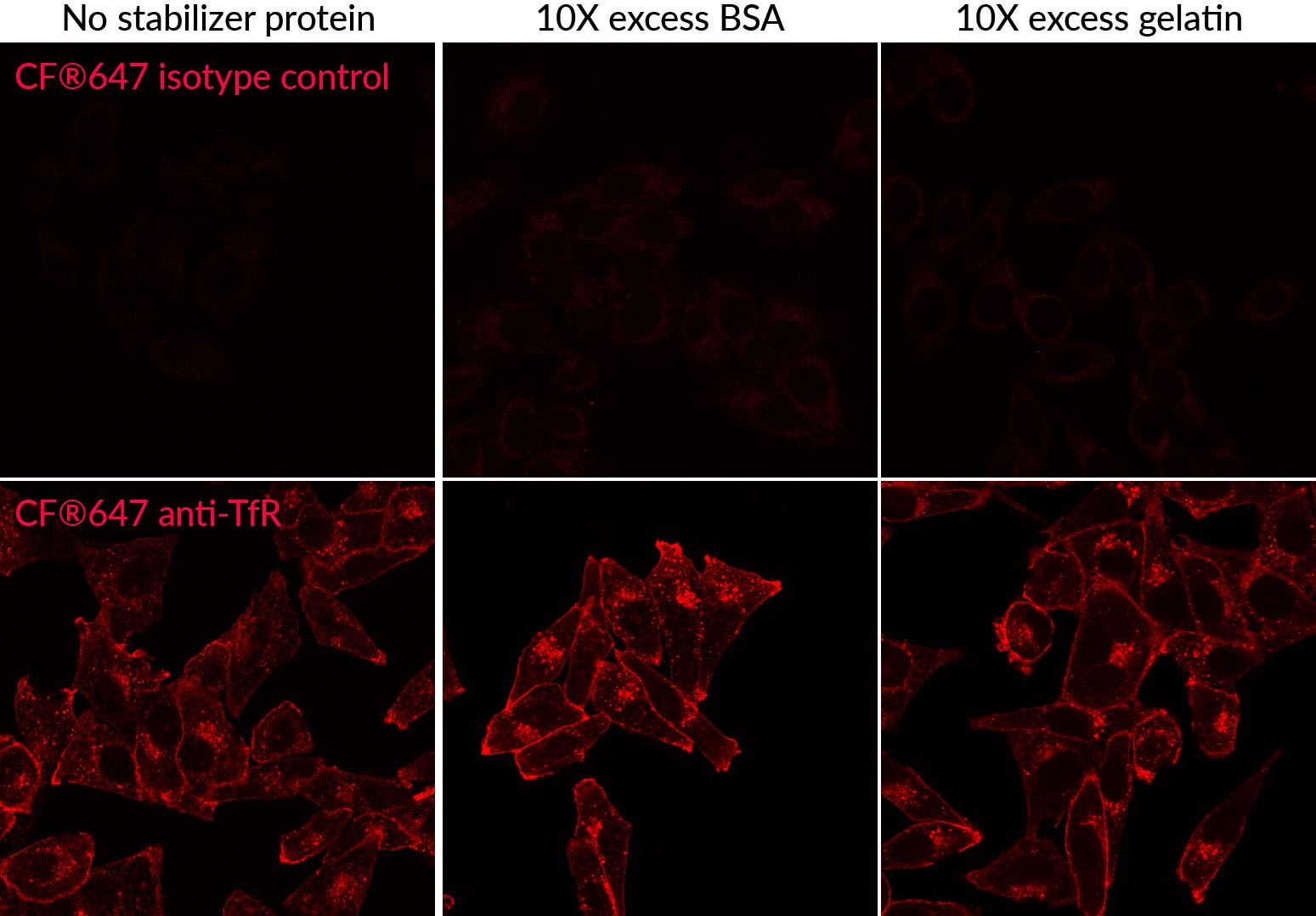
Figure 2. Comparison of Mix-n-Stain™ CF®647 labeling of anti-transferrin antibody (endosome and cell surface marker) in HeLa cells without added protein using the standard protocol compared to using the modified protocol in the presence of 1% BSA or 1% gelatin (10-fold excess) stabilizer protein. Labeling using the modified protocol gives comparable results as the standard protocol, with bright signal and minimal increase in non-specific background.
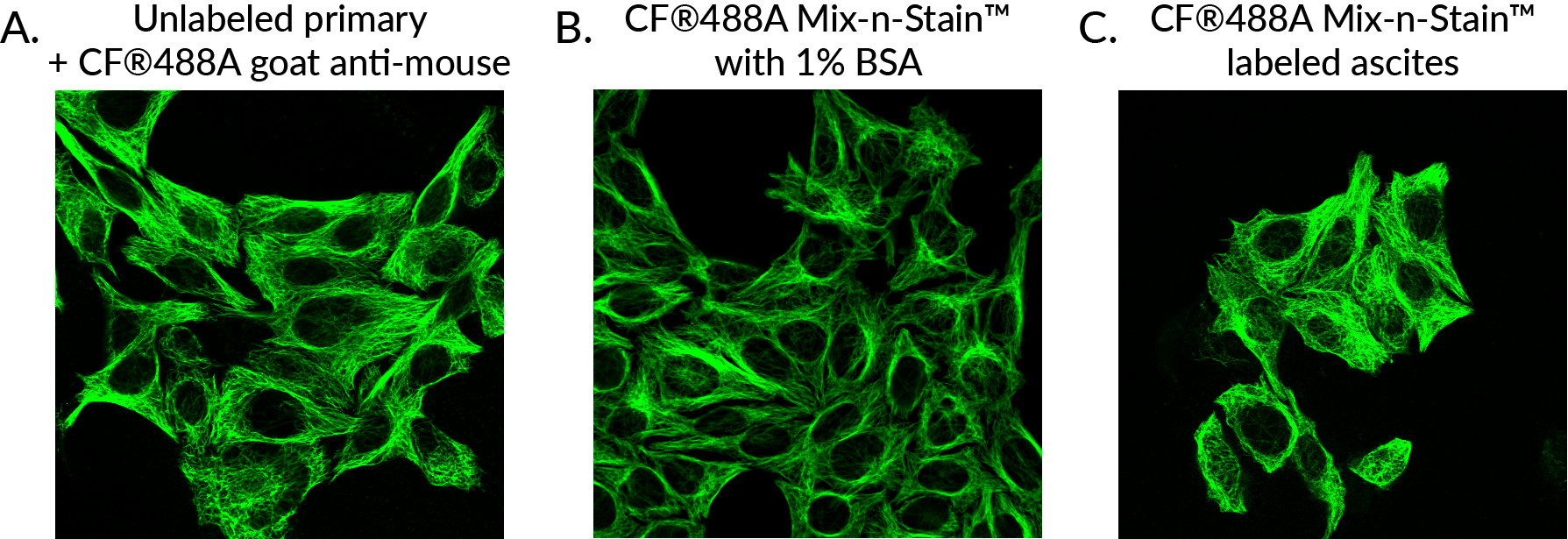
Figure 3. HeLa cells stained with monoclonal mouse anti-tubulin clone DM1A, unconjugated or conjugated using Mix-n-Stain® CF488A antibody labeling using the modified labeling protocol.
Unconjugated primary antibody with (A) CF®488A goat anti-mouse.
(B) CF®488A conjugated antibody labeled in the presence of 1% BSA.
(C) CF®488A conjugated antibody labeled in unpurified ascites fluid.
The Mix-n-Stain™-labeled antibodies performed comparably to unconjugated antibody using indirect detection in immunofluorescence staining of HeLa cells.
A key advantage of Mix-n-Stain™ is flexibility in antibody input. You can label any antibody amount for the kit range using the standard protocol. But for users who wish to conserve precious antibody, using the modified protocol also allows you to label less than 5 ug IgG for small scale experiments. By supplementing small antibody amounts with BSA to meet the recommended protein amount of the Mix-n-Stain™ Kit size, we have seen bright and stable labeling with as little as 1 ug IgG. Figure 4 shows HeLa cells with labeled tubulin using just 1 ug antibody that was conjugated using a 20-50 ug sized CF®488A Mix-n-Stain™ supplemented with 50 ug BSA. A high ratio of BSA:IgG was used in this experiment to demonstrate the tolerance of Mix-n-Stain™ labeling for excess BSA; if you wish to label less than 5 ug of antibody, we recommend adding BSA to bring the total amount of protein in the reaction to 5 ug and using a 5-20 ug size kit. See the Mix-n-Stain™ CF® Dye product information sheet for details.
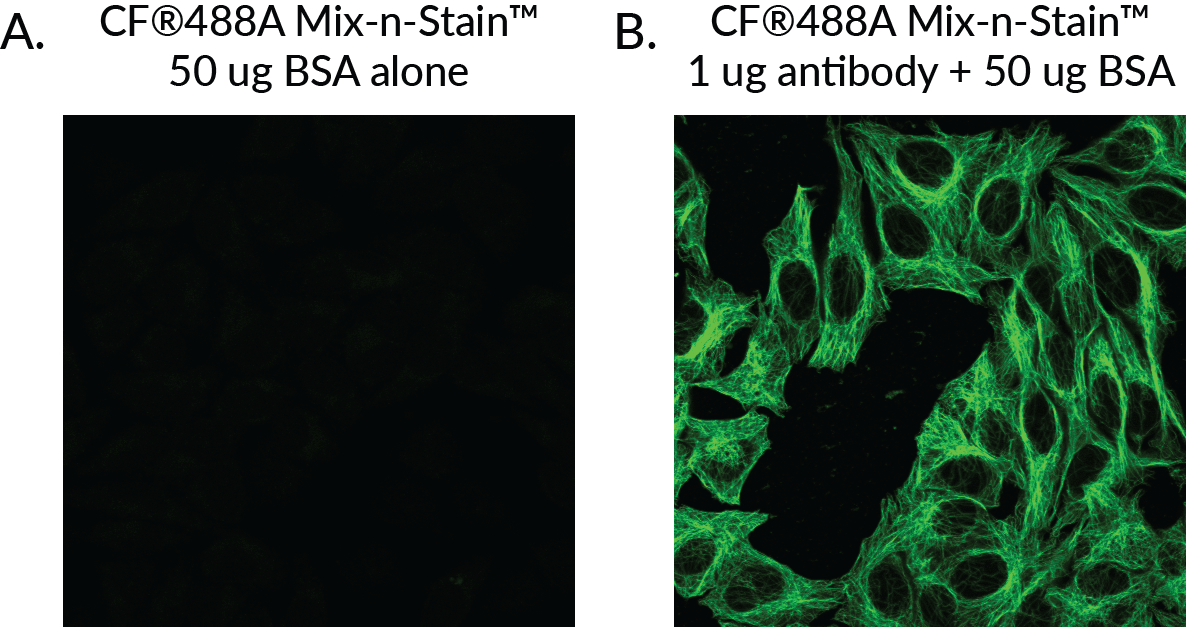
Figure 4. A 20–50 µg-sized CF®488A Mix-n-Stain™ was used to label 50 µg BSA alone (A) or 1 µg mouse anti-tubulin supplemented with 50 µg BSA (B), then the staining reactions were used for immunofluorescence staining of HeLa cells. Results show bright and clear staining of tubulin with negligible background introduced by labeled BSA in the Mix-n-Stain™ reaction.