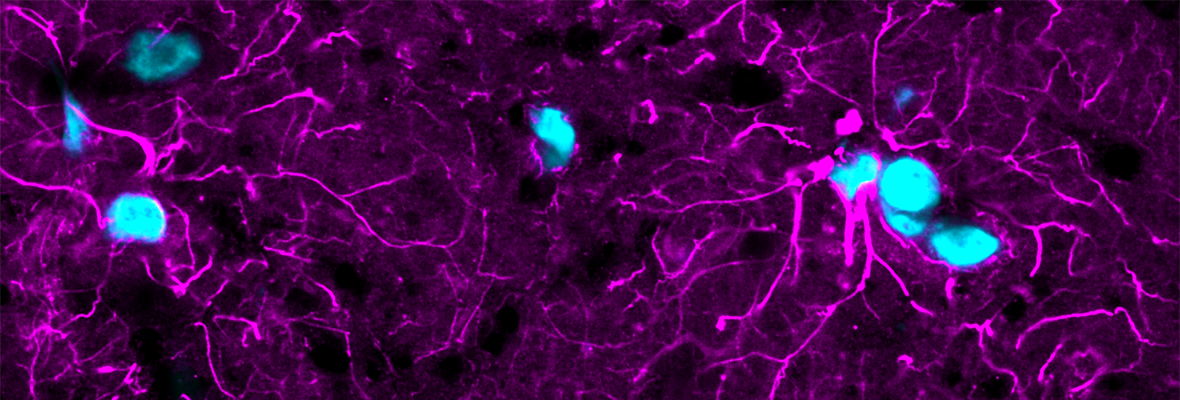
Like a bright sky obscuring the stars, background fluorescence in microscopy makes specific signals harder to see—only the brightest points shine through. Reducing background improves signal-to-noise and enhances image quality, producing better data. Both the intrinsic fluorescence of samples (autofluorescence) and non-specific binding of dyes (background) add to unwanted non-specific signal: background reducers can minimize these.
Reducing Immunofluorescence Background
Non-specific binding of antibodies and negative charges borne by fluorescent dyes increase background. Blocking—typically with normal sera from animals, bovine serum albumin (BSA), or gelatin—competes for non-specific protein-protein interactions and reduces background by minimizing these interactions between the probes and the sample. Animal IgG contained in serum or contaminated BSA can increase background; this can be controlled by careful selection of antibodies and using compatible serum, but becomes problematic in multiplexed immunofluorescence experiments using antibodies from multiple species.
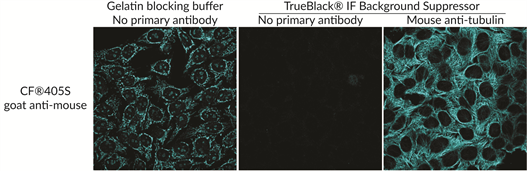
Blocking does not stop charge-mediated interactions, which are pronounced with heavily sulfonated dyes. TrueBlack® IF Background Suppressor System minimizes background from both key sources: non-specific antibody binding and charged fluorescent dyes. The buffer system is compatible with antibodies derived from many species and permeabilizes samples in 10 minutes for one-step prep.
Lipofuscin Fluorescence: What Is It And How Do We Deal With It?
Lipofuscin is a highly fluorescent pigment that is a byproduct of aging and is a type of tissue autofluorescence. Especially pronounced in the brain, retina, and kidneys, its deposition is associated with neurodegeneration (Moreno-García et al., 2018) and has recently been explored as a biomarker for senescent cells (Evangelou & Gorgoulis, 2017; Georgakopoulou et al., 2013). Lipofuscin puncta are fluorescent across the visible spectrum (Schnell et al., 1999) and obscure specific signals.
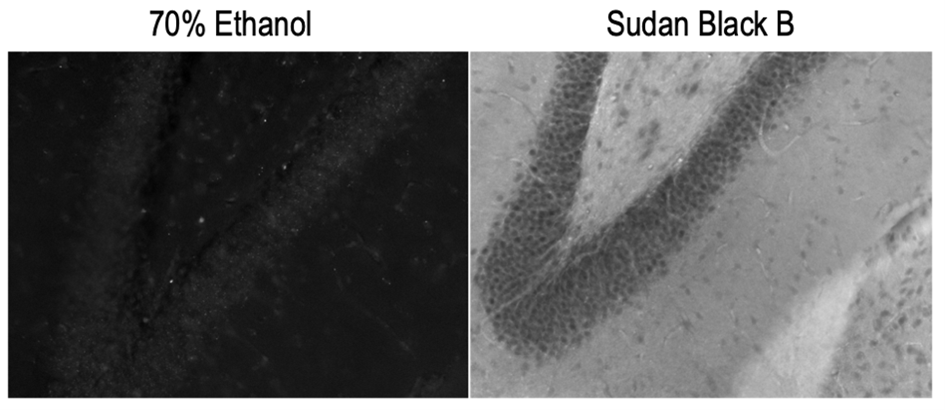
Lipofuscin signal can be handled in two ways: 1) chemical treatment and 2) image processing. Sudan Black B has been the most reliable chemical method for quenching lipofuscin fluorescence (Romijn et al., 1999; Schnell et al., 1999), but it is not without drawbacks. Sudan Black B is fluorescent in the red and far-red range, so its use renders the far-red channel unusable. It also requires an ethanol solvent, which may not be compatible with all staining protocols.
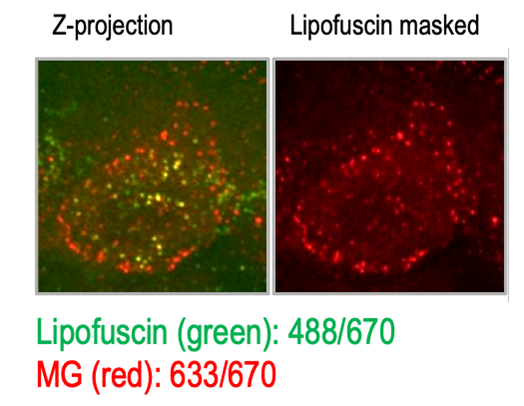
In my past research, I used malachite green—a far-red fluorophore—to label endogenous BK channels (Pratt et al., 2017). Because Sudan Black treatment was not an option, I digitally masked lipofuscin fluorescence identified by its broad fluorescence profile. I was fortunate that my signal of interest was primarily at the plasma membrane and lipofuscin was perinuclear. Newer spectral scanning instruments can remove signal from lipofuscin and other sources of autofluorescence digitally based on their emission spectrum signature; however, not all researchers have access to this option.
Enter TrueBlack® Plus, which quenches lipofuscin autofluorescence without increasing far-red background—allowing improved stains in lipofuscin-laden tissue (and would have helped me immensely in graduate school). Furthermore, TrueBlack® Plus is stable in aqueous solution, making it more versatile than Sudan Black.
Other Applications for Quenching
Western blotting also benefits from reduced background; the same chemical and physical principles cause background in fluorescent western blots as they do in immunofluorescence. Fluorescent detection of antibodies in the western blot format allows for multiplexing on single blots. The TrueBlack® Western Blotting Blocking Buffer Kit is more effective than BSA, gelatin, or casein at improving specificity and reducing non-specific binding to membranes, allowing you to see weaker signals.
References
- Evangelou, K., & Gorgoulis, V. G. (2017). Sudan Black B, The Specific Histochemical Stain for Lipofuscin: A Novel Method to Detect Senescent Cells. Methods in Molecular Biology (Clifton, N.J.), 1534, 111–119. https://doi.org/10.1007/978-1-4939-6670-7_10
- Georgakopoulou, E. A., Tsimaratou, K., Evangelou, K., Fernandez Marcos, P. J., Zoumpourlis, V., Trougakos, I. P., Kletsas, D., Bartek, J., Serrano, M., & Gorgoulis, V. G. (2013). Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging, 5(1), 37–50. https://doi.org/10.18632/aging.100527
- Moreno-García, A., Kun, A., Calero, O., Medina, M., & Calero, M. (2018). An Overview of the Role of Lipofuscin in Age-Related Neurodegeneration. Frontiers in Neuroscience, 12. https://doi.org/10.3389/fnins.2018.00464
- Pratt, C. P., Kuljis, D. A., Homanics, G. E., He, J., Kolodieznyi, D., Dudem, S., Hollywood, M. A., Barth, A. L., & Bruchez, M. P. (2017). Tagging of Endogenous BK Channels with a Fluorogen-Activating Peptide Reveals β4-Mediated Control of Channel Clustering in Cerebellum. Frontiers in Cellular Neuroscience, 11. https://doi.org/10.3389/fncel.2017.00337
- Romijn, H. J., van Uum, J. F. M., Breedijk, I., Emmering, J., Radu, I., & Pool, C. W. (1999). Double Immunolabeling of Neuropeptides in the Human Hypothalamus as Analyzed by Confocal Laser Scanning Fluorescence Microscopy. Journal of Histochemistry & Cytochemistry, 47(2), 229–235. https://doi.org/10.1177/002215549904700211
- Schnell, S. A., Staines, W. A., & Wessendorf, M. W. (1999). Reduction of lipofuscin-like autofluorescence in fluorescently labeled tissue. Journal of Histochemistry & Cytochemistry, 47(6), 719–730. https://doi.org/10.1177/002215549904700601