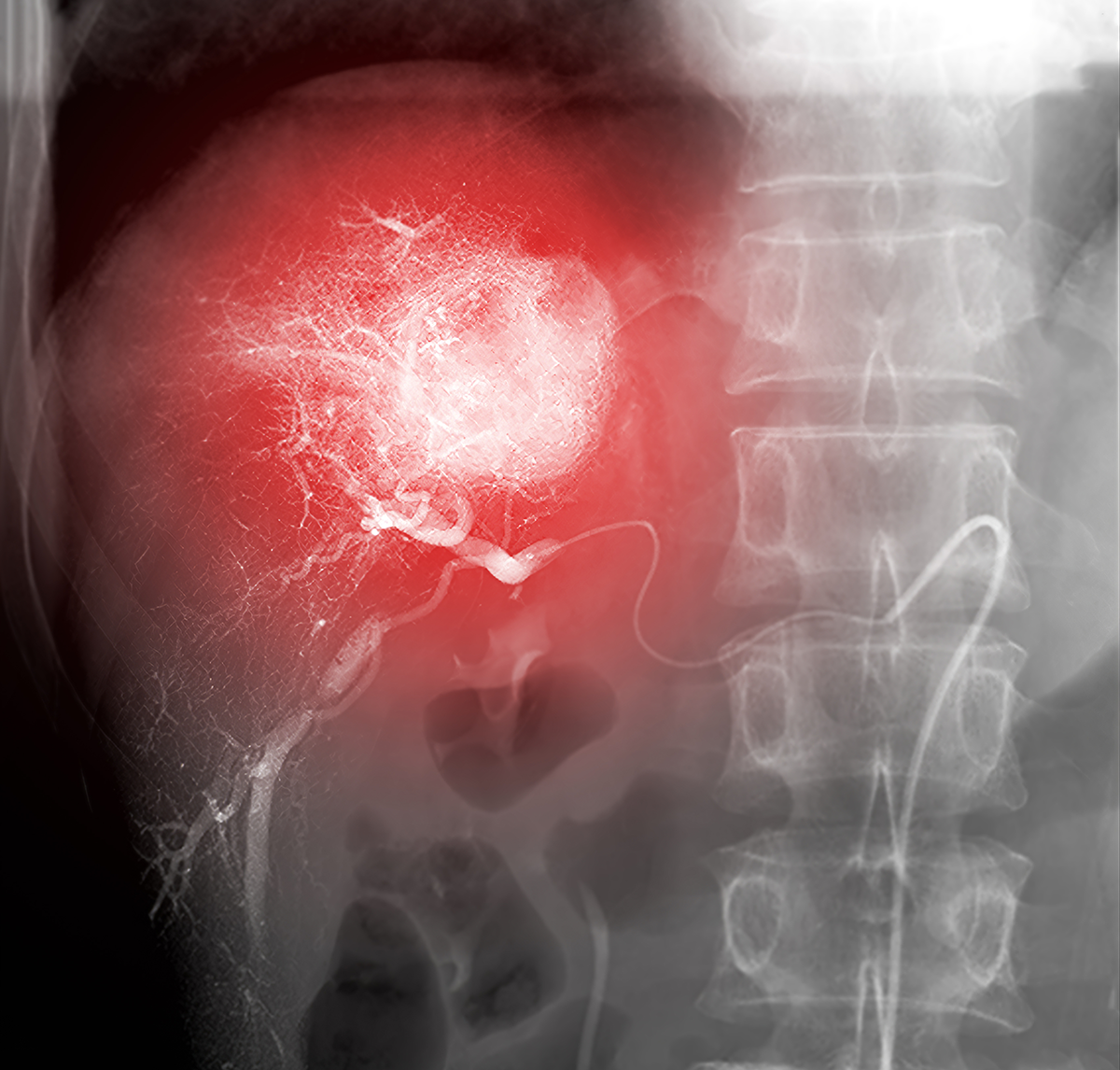
Go Long to See Deeper
Imaging in visible light hits a wall when it comes to imaging in tissue for two main reasons: visible light is absorbed and scattered by tissue and tissue autofluorescence occurs chiefly in the visible spectrum. Both factors limit the usefulness of visible light for looking deep in tissue. Moving into the far red and near-infrared (NIR) region of the spectrum allows light to penetrate deeper into tissue with less absorption, scattering, and autofluorescence, improving signal-to-noise and depth of imaging. NIR spans from 650-2000 nm and contains two spectral “windows” for biologic imaging where water absorption is minimal: NIR-I ranges from 650-900 and NIR-II ranges from 1000-1300, overlapping with short-wave IR (SWIR), which spans 1000-2000 nm. NIR and SWIR imaging are not without drawbacks: spatial resolution decreases with longer wavelengths of light and reduced but present scattering can still degrade image quality. Fortunately, advances in imaging technology including adaptive optics and deep learning can mitigate these limitations.1–5
Clinical Applications of NIR Imaging
Conventional medical scans like ultrasound, magnetic resonance imaging (MRI), computed tomography (CT) or positron emission tomography (PET) scans lack cellular resolution; thus, identifying tumor locations and margins in practice remains a large challenge with these technologies. On the other hand, optical imaging using NIR fluorescent dyes can visualize individual cells. While some NIR dyes preferentially stain tumors over untransformed tissue through mechanisms that aren’t clearly understood, targeted NIR probes—NIR dyes conjugated to an antibody or ligand—can be used to visualize and define tumor margins for surgical excision.3–7
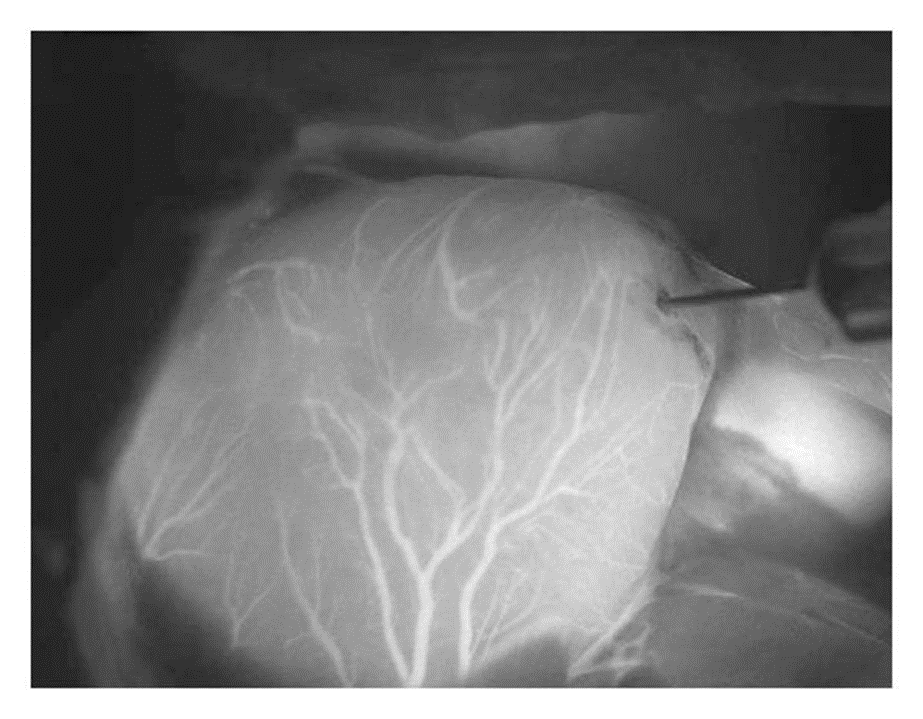
Indocyanine green (ICG), an FDA-approved dye, is useful for imaging vasculature in several organs. ICG is used in practice for coronary surgeries, liver laparoscopy, visualizing infant cerebral blood flow, and is being explored for lymph node mapping in oncology.8,9
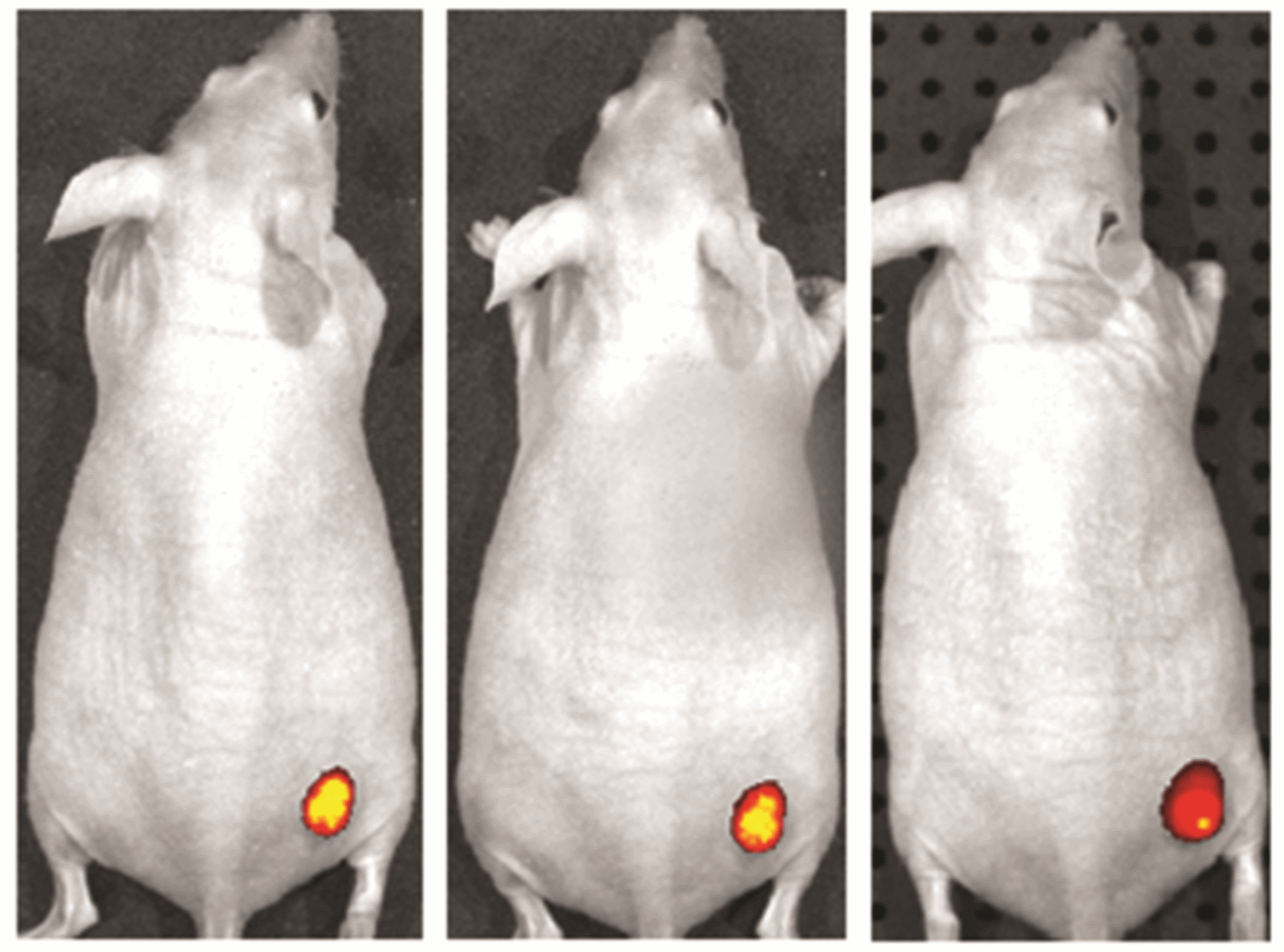
Dyes for NIR
A plethora of probes for NIR imaging have been developed, including organic dyes such as ICG and Biotium’s long wavelength CF® Dyes; nanoparticles such as quantum dots, nanodiamonds, and carbon nanotubes; fluorescent proteins; and bioluminescent protein-substrate complexes. Most NIR organic dyes emit in the NIR-I range, with few emitting in NIR-II and SWIR, which provide even better signal to noise in biological tissues. Most NIR-II emitters are nanoparticles that are poorly biocompatible, containing cytotoxic materials such as heavy metals. Small organic dyes such as near-IR CF® Dyes can penetrate into tissues easily and have a linear response to illumination; the NIR spectral region offers low tissue autofluorescence, producing high sensitivity.1,3,5,7,9
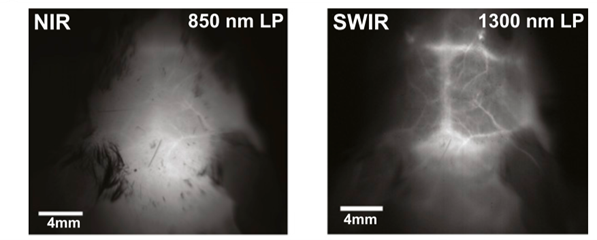
Future Developments
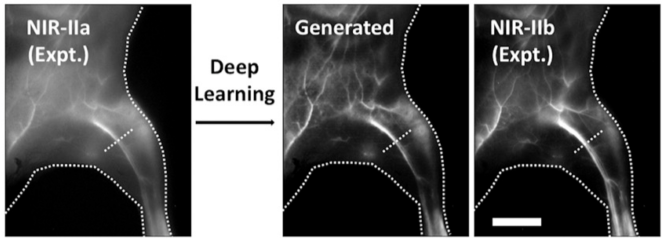
While NIR imaging provides unmatched in vivo imaging capability, there are some remaining limitations: high-resolution imaging at depth still has some scattering, which blurs the image despite light being able to penetrate. This is improved by imaging in NIR-II and SWIR; however, most organic dyes emit in NIR-I. Advances in deep learning and generative adversarial networks (GANs) have made it possible to translate NIR-I images to NIR-II quality with sufficient model training.1
Another developing technology that can improve image quality is adaptive optics. Adaptive optics were originally developed for astronomy to counter distortions by the atmosphere, but the principle can be used in biologic imaging to counteract the image produced by scattering in the tissue, even permitting super-resolution imaging at depth.2
For clinical use, several new NIR dyes have been undergoing clinical trials for use in human surgery and photodynamic therapies, largely in oncology. Biocompatible materials will lead the way in NIR dye development, both in and out of the clinic. The advantages of NIR in basic biological research are also gaining traction as volumetric microscopic imaging techniques such as light sheet become more common. Imaging in NIR-II and SWIR is also expected to become more common, as the InGaAs (indium gallium arsenide) sensors required to detect long wavelength photons become more commonplace.3,4,7,10
References
- Ma Z, Wang F, Wang W, Zhong Y, Dai H. Deep learning for in vivo near-infrared imaging. PNAS. 2021;118(1). doi:10.1073/pnas.2021446118
- Sahu P, Mazumder N. Improving the Way We See: Adaptive Optics Based Optical Microscopy for Deep-Tissue Imaging. Frontiers in Physics. 2021;9:138. doi:10.3389/fphy.2021.654868
- Tiwari DK, Tiwari M, Jin T. Near-infrared fluorescent protein and bioluminescence-based probes for high-resolution in vivo optical imaging. Mater Adv. 2020;1(5):967-987. doi:10.1039/D0MA00273A
- Zhang RR, Schroeder AB, Grudzinski JJ, et al. Beyond the margins: real-time detection of cancer using targeted fluorophores. Nat Rev Clin Oncol. 2017;14(6):347-364. doi:10.1038/nrclinonc.2016.212
- Zhang X, Bloch S, Akers W, Achilefu S. Near-infrared Molecular Probes for In Vivo Imaging. Curr Protoc Cytom. 2012;CHAPTER:Unit12.27. doi:10.1002/0471142956.cy1227s60
- Rumyantsev KA, Turoverov KK, Verkhusha V. Near-infrared bioluminescent proteins for two-color multimodal imaging. Sci rep. 2016;6:36588. doi:10.1038/srep36588
- Yi X, Wang F, Qin W, Yang X, Yuan J. Near-infrared fluorescent probes in cancer imaging and therapy: an emerging field. IJN. 2014;9(1):1347-1365. doi:10.2147/IJN.S60206
- Alander JT, Kaartinen I, Laakso A, et al. A Review of Indocyanine Green Fluorescent Imaging in Surgery. International Journal of Biomedical Imaging. 2012;2012:e940585. doi:10.1155/2012/940585
- Carr JA, Franke D, Caram JR, et al. Shortwave infrared fluorescence imaging with the clinically approved near-infrared dye indocyanine green. PNAS. 2018;115(17):4465-4470. doi:10.1073/pnas.1718917115
- Smith AM, Mancini MC, Nie S. Second window for in vivo imaging. Nature Nanotech. 2009;4(11):710-711. doi:10.1038/nnano.2009.326