Understanding microbes and their ecology is crucial for modern medicine and functioning society. Microbes cause disease, but are also vital components of a healthy gut and skin microbiome. Microbes fix nitrogen for plants, produce oxygen in marine environments, facilitate rusting and infrastructure breakdown, and come with us to space. By better understanding microbial diversity and their interactions with hosts and each other, we can reduce infections and food-borne illness, improve wastewater treatment and irrigation, and better human health (Emerson et al., 2017).
Culture viability—growing microbes in controlled media and counting colonies—has been the standard method for identifying and quantifying living microbes, but culture growth takes time. Worse still, certain organisms cannot be cultured with current technology. Cultures are ideal when looking for known, culturable species of interest, but these viable but non-culturable species limits their utility for ecological and complex microbiome studies. Culture-free methods eliminate these limitations but require molecular measures of viability.
Which are the dead ones?
With a few exceptions, the cell membrane breaks down and becomes permeable in dying and dead cells. Propidium iodide (PI)—a fluorescent dye commonly used in microscopy and flow cytometry—enters cells with compromised membranes and intercalates into the DNA, activating fluorescence for optical detection. PI, however, can show “false labeling” under certain conditions, such as with adherent biofilms or with extracellular debris, overestimating the population of dead cells (Rosenberg et al., 2019). PI performance is also species and growth phase-dependent: one study found that up to 10% of PI-labeled Gram-negative Sphingomonas species and 60% Gram-positive M. fredriksbergense were actually viable (Shi et al., 2007). It is unclear if PI false labeling involves cell entry or if fluorescence is extracellular; if the latter, then non-optical methods could circumvent problematic labeling.
Viability PCR and Molecular Approaches
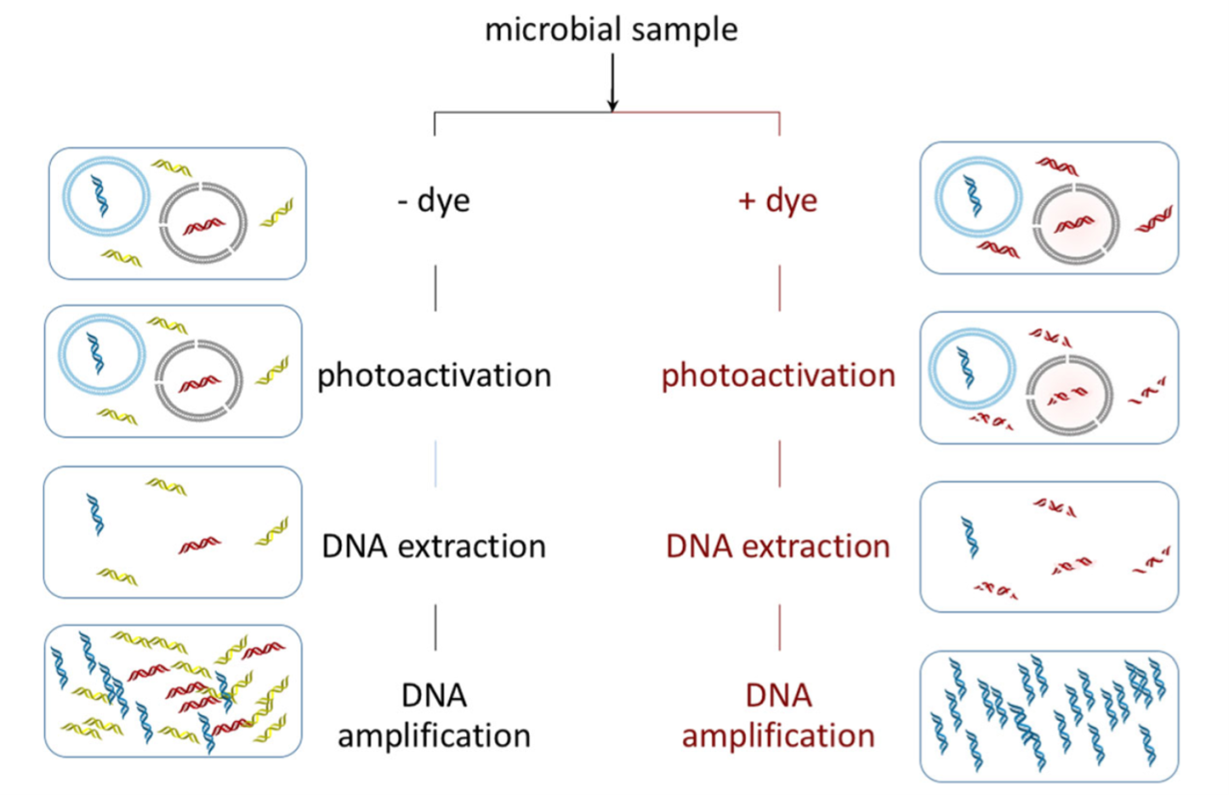
PCR by itself cannot distinguish between DNA from living or dead cells. In 2006, Biotium invented and remains the primary supplier of propidium monoazide (PMA) (Madrid, 2018; Nocker et al., 2006). Like PI, PMA is excluded from living cells but labels DNA in dead or dying cells (Emerson et al., 2017; Nocker et al., 2006). With light activation, PMA bonds covalently to DNA via its azide functional group, rendering DNA less soluble and a poor template for amplification (Emerson et al., 2017; Nocker et al., 2006). Viability PCR (v-PCR) with PMA “blocks” extracellular and dead-cell DNA from amplification in many downstream PCR applications (Emerson et al., 2017).
A Clinical Research Tool
High throughput sequencing and microbial profiling with v-PCR can enhance our understanding of microbial communities and their evolution. Lung infections are a significant cause of complications and mortality in cystic fibrosis (CF) patients (Lyczak et al., 2002). PMA-treated v-PCR and 16S-rRNA gene sequencing of sputum samples from 30 CF patients identified microbes—including potential pathogens—that were missed without PMA treatment (Rogers et al., 2013).
Validating Household Cleaners
At home, our plumbing hosts biofilms that can breed pathogens. Optimizing benzalkonium chloride formulations to kill biofilms isolated from household kitchen drains was made possible with v-PCR (Forbes et al., 2017): simple aqueous solutions had little effect on the drain biofilms, but a complex formulation with additives typical for household cleaners reduced DNA recovery from PMA-treated biofilms, indicating microbial lethality. v-PCR allows us to counter threats to health by better measuring and understanding microbial communities in health and disease.
Surveilling Risks for Food-borne Illness
Waste runoff and contaminated irrigation are sources of foodborne illness worldwide. Beyond bacteria, v-PCR has been used in viability studies of viruses and eukaryotes to improve sanitary practices in agriculture and environmental management (Alonso et al., 2014; López‐Gálvez et al., 2018; Madrid, 2018; Prevost et al., 2016; Randazzo et al., 2016). v-PCR has detected pathogenic protozoan viability (Alonso et al., 2014) and can distinguish between infectious and non-infectious norovirus in water samples (Prevost et al., 2016; Randazzo et al., 2016). No country is immune to foodborne illness from many pathogens, and the ability to evaluate and improve sanitary measures at scale through viability testing remains as crucial as ever.
A Microbial Space Odyssey
As we reach for the stars and plan interplanetary missions, we need to understand the microbial environments that form in space to protect astronauts and future spacefarers. Clean rooms on Earth are often used to simulate space conditions; samples taken from the International Space Station (ISS) and cleanrooms at the Jet Propulsion Lab revealed substantial differences in their microbiomes (Checinska et al., 2015). Airborne and settled debris in the ISS had only 1.7% and 2.7% viable population, respectively, and the living constituents were enriched in Actinobacteria—human skin commensals. Being desiccation-resistant, actinobacteria were especially dominant in the ISS HEPA filtration units. Because of the vanishingly small proportion of surviving microbes, these key differences between space and Earthbound environments would not have been possible without PMA to block material contributions from non-living sources.
Future Directions
v-PCR is a powerful technique that has grown immensely since the late-2000s (Madrid, 2018); its versatility shows strength in viability and diversity studies with myriad applications. The strengths of PCR in identifying bacterial, protozoan, viral, and even archaeal constituents have been limited only by selectivity for live cells; PMA entered the scene as a more selective replacement for ethidium monoazide (Nocker et al., 2006). Like PI, PMA’s performance varies by species and may suffer when used in complex, multispecies environments (Wang et al., 2021). PMAxx™, Biotium’s next-generation v-PCR dye, has improved selectivity and dead cell permeability compared to PMA and will lead the way in coming v-PCR applications (López‐Gálvez et al., 2018; Madrid, 2018; Randazzo et al., 2016)!
References
- Alonso, J. L., Amorós, I., & Guy, R. A. (2014). Quantification of viable Giardia cysts and Cryptosporidium oocysts in wastewater using propidium monoazide quantitative real-time PCR. Parasitology Research, 113(7), 2671–2678. https://doi.org/10.1007/s00436-014-3922-9
- Checinska, A., Probst, A. J., Vaishampayan, P., White, J. R., Kumar, D., Stepanov, V. G., Fox, G. E., Nilsson, H. R., Pierson, D. L., Perry, J., & Venkateswaran, K. (2015). Microbiomes of the dust particles collected from the International Space Station and Spacecraft Assembly Facilities. Microbiome, 3(1), 50. https://doi.org/10.1186/s40168-015-0116-3
- Emerson, J. B., Adams, R. I., Román, C. M. B., Brooks, B., Coil, D. A., Dahlhausen, K., Ganz, H. H., Hartmann, E. M., Hsu, T., Justice, N. B., Paulino-Lima, I. G., Luongo, J. C., Lymperopoulou, D. S., Gomez-Silvan, C., Rothschild-Mancinelli, B., Balk, M., Huttenhower, C., Nocker, A., Vaishampayan, P., & Rothschild, L. J. (2017). Schrödinger’s microbes: Tools for distinguishing the living from the dead in microbial ecosystems. Microbiome, 5(1), 86. https://doi.org/10.1186/s40168-017-0285-3
- Forbes, S., Cowley, N., Humphreys, G., Mistry, H., Amézquita, A., & McBain, A. J. (2017). Formulation of Biocides Increases Antimicrobial Potency and Mitigates the Enrichment of Nonsusceptible Bacteria in Multispecies Biofilms. Applied and Environmental Microbiology, 83(7). https://doi.org/10.1128/AEM.03054-16
- López‐Gálvez, F., Randazzo, W., Vásquez, A., Sánchez, G., Decol, L. T., Aznar, R., Gil, M. I., & Allende, A. (2018). Irrigating Lettuce with Wastewater Effluent: Does Disinfection with Chlorine Dioxide Inactivate Viruses? Journal of Environmental Quality, 47(5), 1139–1145. https://doi.org/10.2134/jeq2017.12.0485
- Lyczak, J. B., Cannon, C. L., & Pier, G. B. (2002). Lung Infections Associated with Cystic Fibrosis. Clinical Microbiology Reviews, 15(2), 194–222. https://doi.org/10.1128/CMR.15.2.194-222.2002
- Madrid, A. (2018, June 1). 12 years later: Viability PCR using PMA and PMAxxTM dyes. The Full Spectrum. https://biotium.com/blog/12-years-later-viability-pcr-using-pma-and-pmaxx-dyes/
- Nocker, A., Cheung, C.-Y., & Camper, A. K. (2006). Comparison of propidium monoazide with ethidium monoazide for differentiation of live vs. Dead bacteria by selective removal of DNA from dead cells. Journal of Microbiological Methods, 67(2), 310–320. https://doi.org/10.1016/j.mimet.2006.04.015
- Prevost, B., Goulet, M., Lucas, F. S., Joyeux, M., Moulin, L., & Wurtzer, S. (2016). Viral persistence in surface and drinking water: Suitability of PCR pre-treatment with intercalating dyes. Water Research, 91, 68–76. https://doi.org/10.1016/j.watres.2015.12.049
- Randazzo, W., López-Gálvez, F., Allende, A., Aznar, R., & Sánchez, G. (2016). Evaluation of viability PCR performance for assessing norovirus infectivity in fresh-cut vegetables and irrigation water. International Journal of Food Microbiology, 229, 1–6. https://doi.org/10.1016/j.ijfoodmicro.2016.04.010
- Rogers, G. B., Cuthbertson, L., Hoffman, L. R., Wing, P. A., Pope, C., Hooftman, D. A. P., Lilley, A. K., Oliver, A., Carroll, M. P., Bruce, K. D., & van der Gast, C. J. (2013). Reducing bias in bacterial community analysis of lower respiratory infections. The ISME Journal, 7(4), 697–706. https://doi.org/10.1038/ismej.2012.145
- Rosenberg, M., Azevedo, N. F., & Ivask, A. (2019). Propidium iodide staining underestimates viability of adherent bacterial cells. Scientific Reports, 9(1), 6483. https://doi.org/10.1038/s41598-019-42906-3
- Shi, L., Günther, S., Hübschmann, T., Wick, L. Y., Harms, H., & Müller, S. (2007). Limits of propidium iodide as a cell viability indicator for environmental bacteria. Cytometry Part A, 71A(8), 592–598. https://doi.org/10.1002/cyto.a.20402
- Wang, Y., Yan, Y., Thompson, K. N., Bae, S., Accorsi, E. K., Zhang, Y., Shen, J., Vlamakis, H., Hartmann, E. M., & Huttenhower, C. (2021). Whole microbial community viability is not quantitatively reflected by propidium monoazide sequencing approach. Microbiome, 9(1), 17. https://doi.org/10.1186/s40168-020-00961-3