Tumors in vivo are composed of a complex milieu of tumor cells, immune cells, mesenchymal cells and extracellular matrix, collectively known as the tumor microenvironment. During the development of potential new cancer therapies, it is ideal to test the drugs on a system as close to an actual tumor microenvironment as possible, since some compounds that seem promising in a basic 2-D cell culture model are not effective in an actual tumor. In addition, it is desirable to test actual patient tumor samples to try to predict whether that particular patient will respond to a certain treatment.
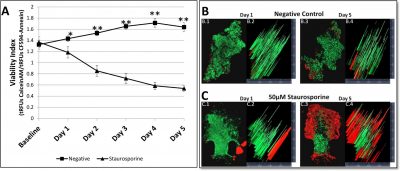
In a recent issue of PLoS ONE, Holton et al. describe the development of a device to trap fine needle aspirate biopsy (FNAB) samples in a microfluidics system, and then monitor various treatments for apoptosis and viability over time. They validated their device by treating tumor samples with a small-molecule drug (doxorubicin), large molecules (dye-labeled antibodies), and the PKC inhibitor staurosporine. For each treatment, cell viability was measured with Calcein AM, and apoptosis was measured by CF™594-Annexin V staining. The ratio of these two stains was used to calculate a viability index for each sample.
Penetration efficiency in the system was determined first by perfusing FNAB samples with doxorubicin. Fluorescence of the drug showed that the sample was uniformly stained, and that apoptosis appeared to have been initiated, as expected. Next fluorescent antibodies were perfused, and showed what appeared to be specific staining throughout the samples. Finally, to demonstrate the usefulness of the system for drug studies on tumor biopsies, they compared 18 control FNAB samples to 18 samples treated with staurosporine over 5 days and measured the viability index as described above (they noted that while the Calcein AM signal photobleached after 5 days, CF™594-Annexin V remained bright after 10 days in the cells). Already after day 1, the viability of the staurosporine-treated samples had decreased, and by 5 days more than 70% of the tumor cells were apoptotic.
The device described here was validated for it’s ability to treat fine needle aspirate biopsy samples with drugs and measure their effects over time using fluorescent markers. It could be a useful tool in the future for personalized cancer therapy.
To read the original article, click here.
Holton AB, Sinatra FL, Kreahling J, Conway AJ, Landis DA, Altiok S (2017) Microfluidic Biopsy Trapping Device for the Real-Time Monitoring of Tumor Microenvironment. PLoS ONE 12(1): e0169797. doi:10.1371/journal.pone.0169797
Biotium has an extensive list of CF™ dye bioconjugates, including Annexin V. For more information on our CF™ dyes, click here.

