Cell migration through 3D extracellular matrix (ECM) is essential during tissue development, wound healing, immune cell trafficking, and during cancer invasion and metastasis. The cyclical multi-step migration process consists of: 1) actin-dependent pseudopod protrusion of the cell’s leading edge; 2) integrin-mediated adhesion; 3) cleavage of ECM elements by cell surface enzymes; 4) actomyosin-dependent contraction of the cell to increase longitudinal tension; and 5) movement of the cell. The deformability of the fairly rigid nucleus has also been shown to act as a determining factor during the passage of cells through tight spaces in the ECM. Consequently, there is growing interest in researching how the nucleus deforms to fit through small gaps despite its stiffness. Nuclear protein lamin A/C has been shown to contribute to nuclear stiffness and can limit unobstructed cell migration in vitro. However, its role in nuclear deformation and passage through obstacle-laden ECM environments was not clear, prompting the study presented here.
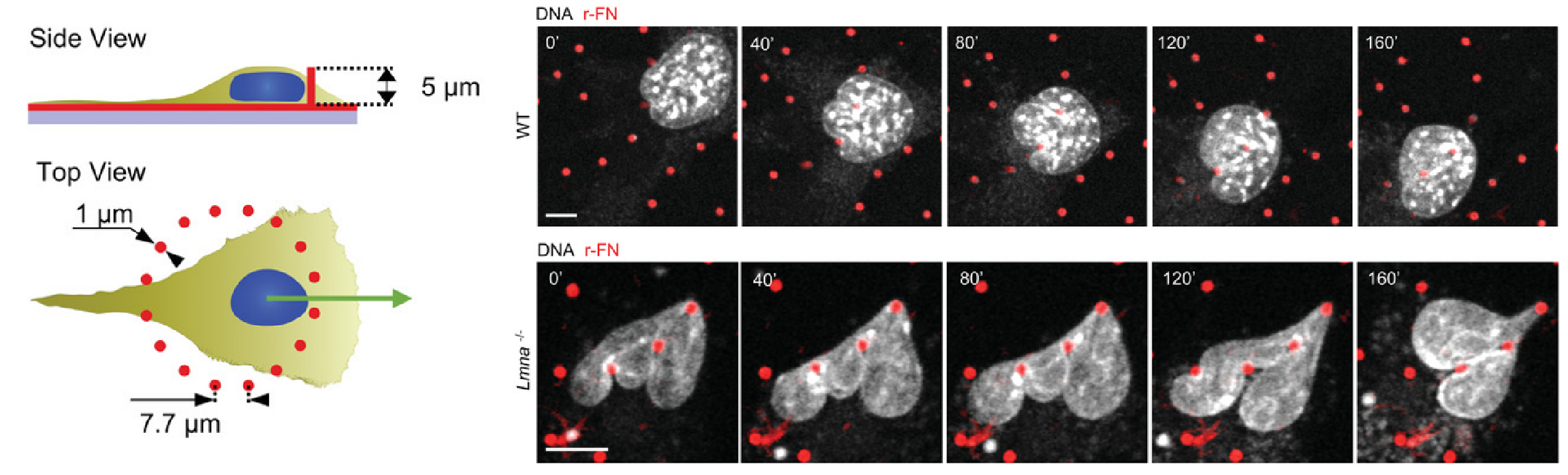
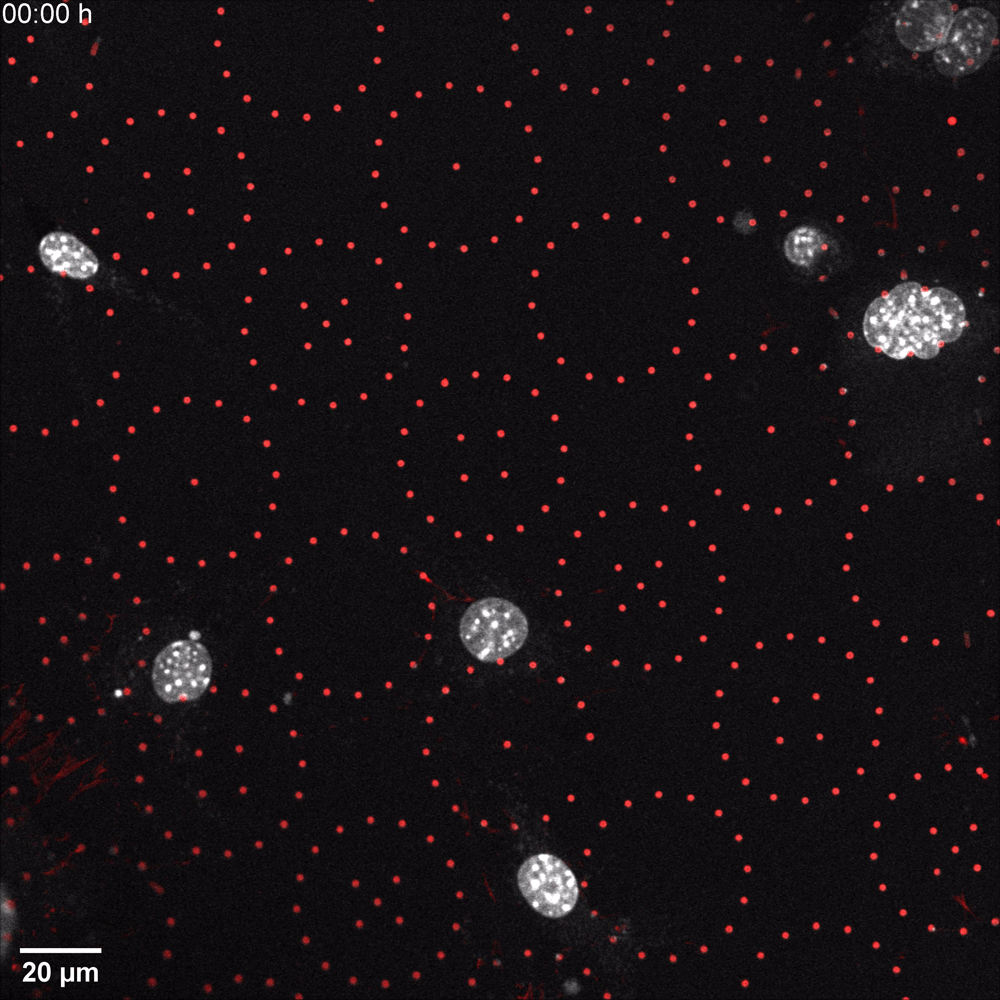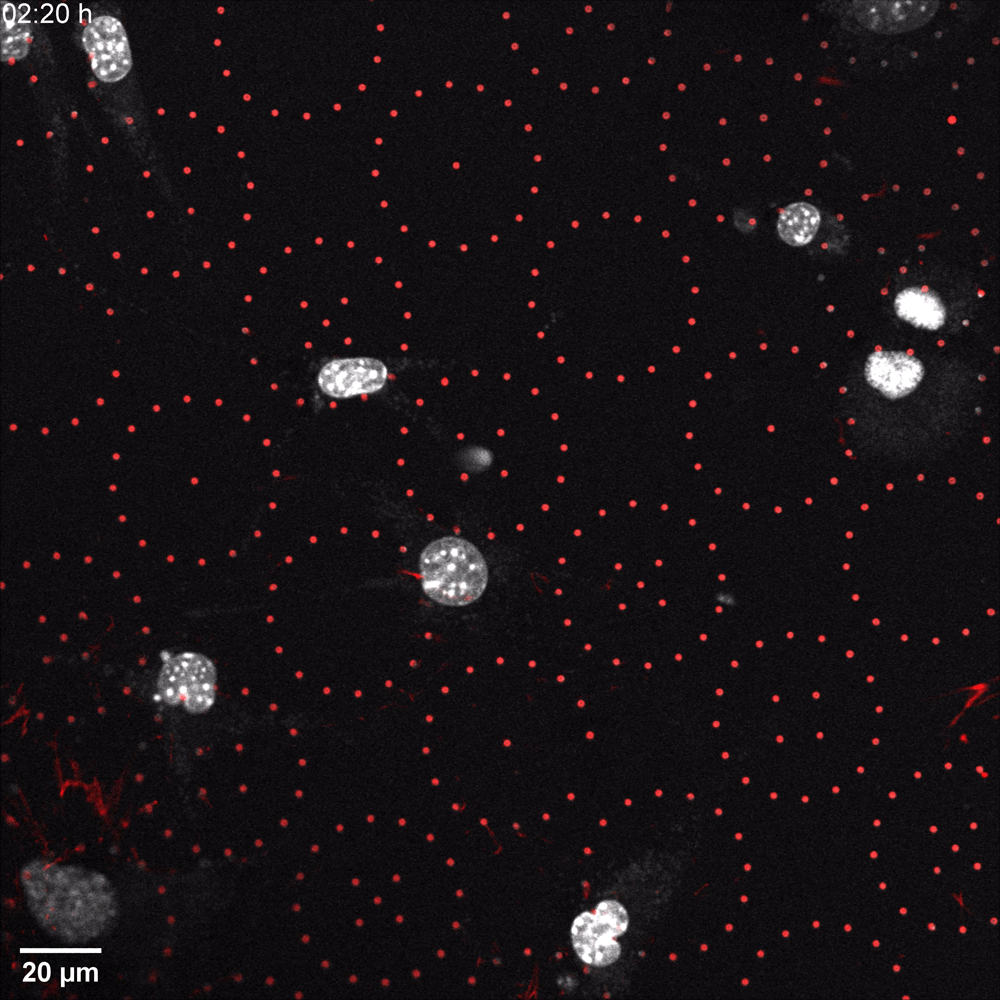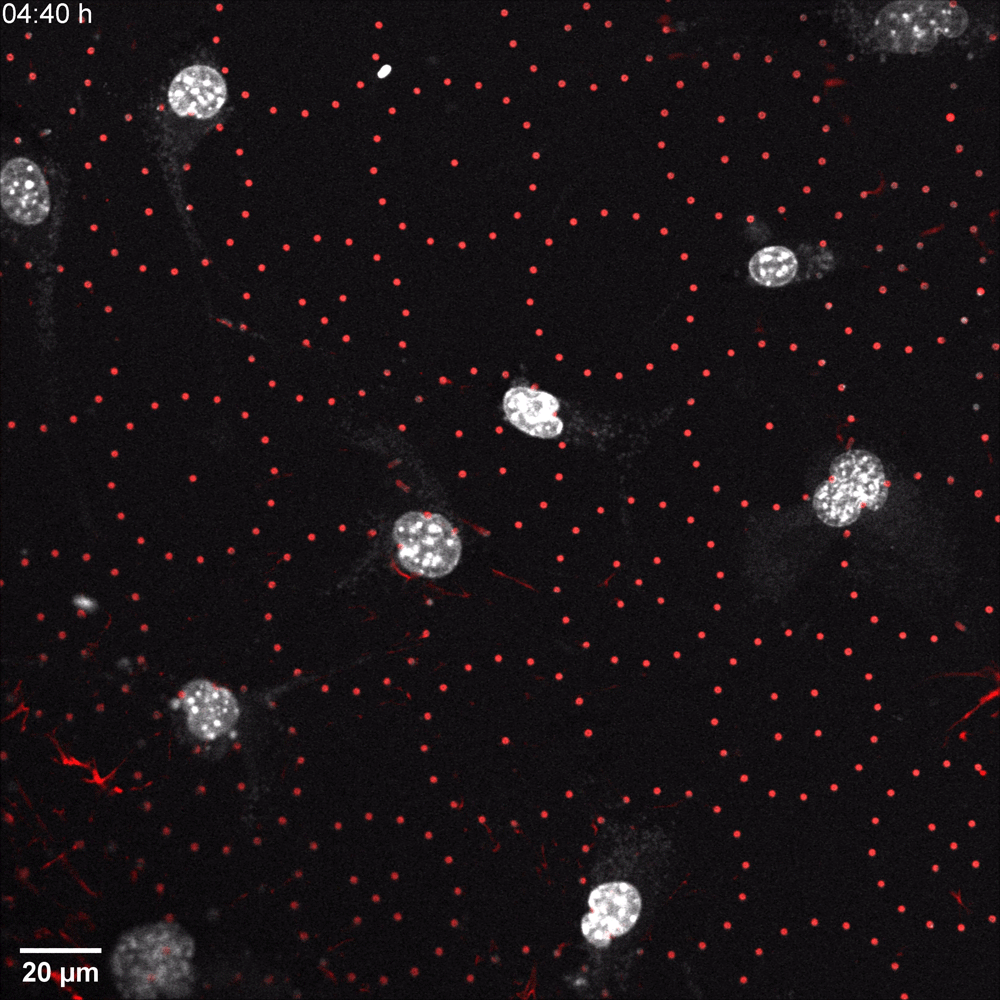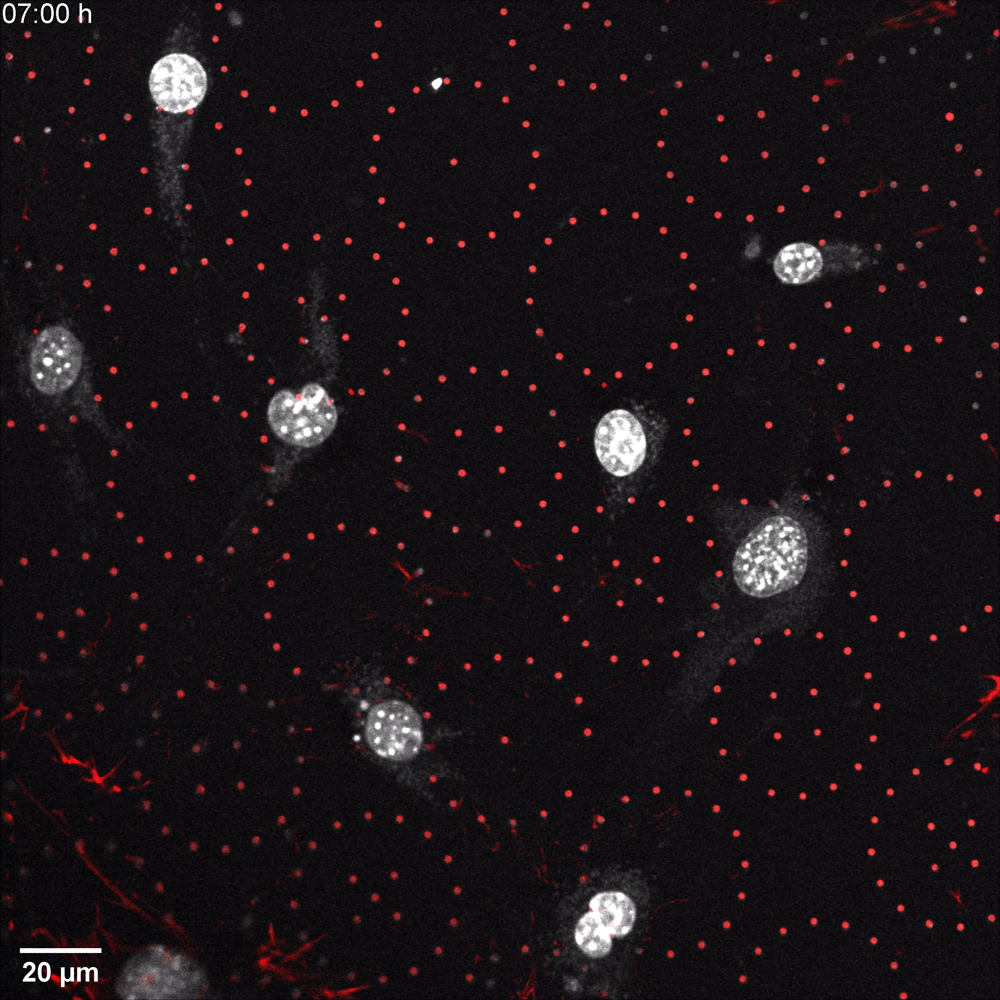
In a recent microscopy-driven publication with striking images and videos, Katiyar et al. examined the role of lamin A/C in cell migration through discrete, closely spaced obstacles designed with physical properties simulating the ECM. They cultured murine 3T3 fibroblasts or embryonic fibroblasts (MEFs) on an array of polydimethylsiloxane (PDMS) micropost barriers, which were continuous with an underlying PDMS surface (Figure 1, Left). The microposts were designed to be similar in size and rigidity to collagen fiber bundles and were arranged so that the space between adjacent microposts was smaller than typical nuclei in cultured cells. The team then used time-lapse confocal fluorescence imaging (Figure 1 and 2) to examine the nuclear shape in migrating cells as they contorted around microposts under a variety of conditions. In order to test for lamin A/C involvement, they used live cell imaging with Biotium’s NucSpot® Live 650 nuclear stain to study nuclear distortion of migrating MEF cells lacking the gene that encodes lamin A/C (Lmna−/−) versus wild-type MEFs. Live-cell imaging revealed that Lmna−/− nuclei became entangled around microposts and were unable to pass around the microposts. Conversely, wild-type nuclei preserved overall nuclear shape despite forming local invaginations, and were able to pass. The wild-type behavior could be rescued in Lmna−/− cells by inducing the expression of GFP-lamin A/C. Based on their results, the team hypothesized that lamin A/C supports surface tension that balances forces exerted on the nuclear membrane and allows the formation of local invaginations without entangling the nucleus, thereby permitting forward motion in mammalian cells.
Along with our NucSpot® Live Cell Nuclear Stains, Biotium carries a range of other nuclear and organelle stains, abundant primary antibody options, and other reagents for optimizing your immunofluorescence microscopy workflows.
Full Citation
Katiyar, A., Zhang, J., Antani, J. D. et al. The nucleus bypasses obstacles by deforming like a drop with surface tension mediated by lamin A/C. Advanced Science, 9(23), 2201248 (2022). https://doi.org/10.1002/advs.202201248

