Content #1
Content #1
Content #1
Cross-talk between fluorescence channels is an important concern for multi-color imaging experiments. The detection of fluorescence from one dye in multiple detection channels can result in artifactual co-localization of signals. Cross-talk usually occurs because of the overlap of dye excitation and emission spectra, and can be managed by careful matching of dye selections to instrument filters, titration of probes to prevent overly bright signal, and imaging each channel individually to prevent cross-excitation by different laser lines. In any multi-color imaging experiment, single stain controls (separate samples stained with each probe by itself) are crucial for ensuring that cross-talk is minimized.
Another source of cross-talk that researchers may be less familiar with is dye photoconversion. This is a change in the spectral properties of a fluorophore that results from its exposure to light. The commonly used blue fluorescent nuclear dyes DAPI and Hoechst undergo UV-induced photoconversion to form green- and red-emitting fluorophores (Zurek-Biesiada et al. 2013, Karg and Golic 2017). Many microscopists are accustomed to using epifluorescence with UV excitation to view and focus on Hoechst- or DAPI-stained nuclei before viewing fluorescence in other channels. This practice can cause DAPI and Hoechst to fluoresce in both the DAPI channel and the FITC or Cy®3 channels. While it is much less bright than the original blue dye fluorescence, the green nuclear signal from the photoconversion product could interfere with imaging of other fluorescent probes or labeled antibodies, especially for low abundance targets, appearing as spurious nuclear localization. Glycerol-based mounting medium, which often includes DAPI as a counterstain, enhances UV-induced photoconversion.
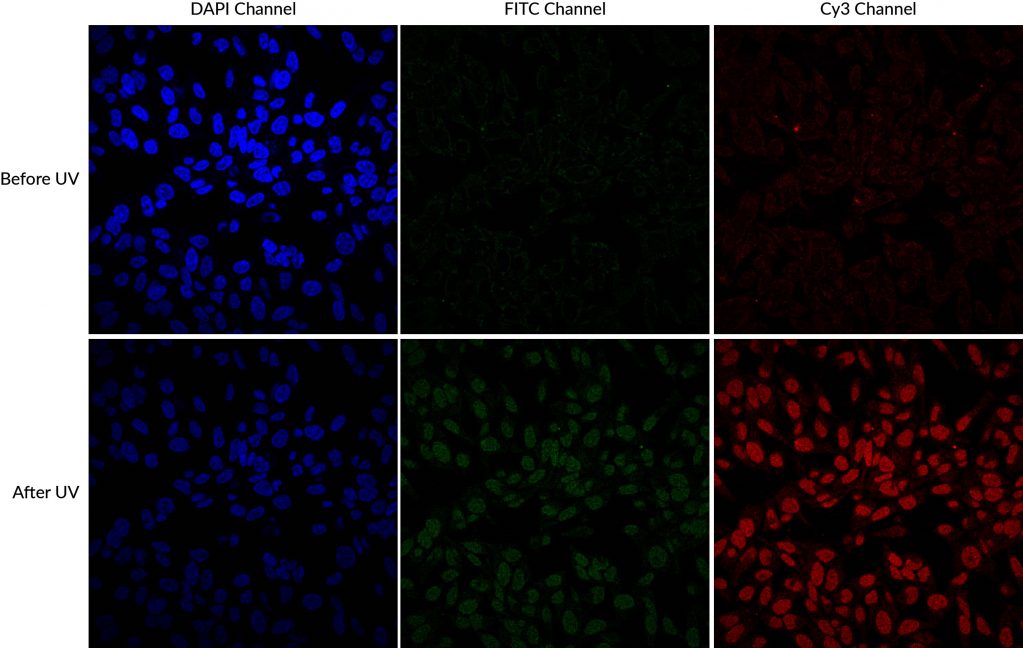
DAPI and Hoechst undergo photoconversion after exposure to UV wavelengths, but not longer excitation wavelengths such as FITC filter cubes or the 405 nm laser line. Once aware of the potential for photoconversion to green fluorescence, it is possible to avoid artifacts through careful imaging practices. One can image green fluorescence before switching to the DAPI channel, or move to an unexposed field of view before imaging the green channel after UV exposure of the sample. Using hardset mounting medium like EverBriteTM Hardset instead of glycerol-based wet-set medium can reduce photoconversion (Roberts et al. 2019). If confocal imaging using the 405 laser line is an option, it is possible to avoid the use of epifluorescence to detect DAPI or Hoechst altogether, which unfortunately sacrifices their utility as aids for finding and focusing on cells. Finally, it is important to include single stain controls for blue nuclear counterstains, especially when detecting low abundance or nuclear targets in other channels.
If photoconversion issues or your experimental design is leading you to avoid using DAPI or Hoechst altogether, Biotium's NucSpot® Nuclear Stains may be a convenient option. These cell-membrane impermeant stains are designed for nuclear specific staining in fixed cells, or selectively staining dead cells in live culture. The stains are available in 7 different colors from green to near-IR for flexible multiplexing. In addition, unlike other green or far-red nucleic acid dyes such as propidium iodide (PI), TOTO®, TO-PRO®, and similar dyes that stain both the nucleus and cytoplasm, NucSpot® Nuclear Stains selectively stain the nucleus in fixed and permeabilized cells without the need for RNase treatment. Biotium also offers NucSpot® Live Cell Nuclear Stains designed for low-toxicity staining for real-time imaging. NucSpot® Live Cell Nuclear Stains are also nuclear specific, compatible with no-wash staining, and include options for FITC or Cy®5 channels.
Alternative far-red nuclear counterstains, such as Biotium's RedDotTM1 and RedDotTM2, avoid the problem of UV photoconversion of DAPI and Hoechst. RedDotTM1 is cell-membrane permeant, and can be used a nuclear stain for live cells, while RedDotTM2 is a counterstain for fixed and permeabilized cells. RedDotTM2 is cell-membrane impermeant, and also can be used for selective staining of dead cells. One disadvantage of RedDotTM1 is that it is more cytotoxic than the commonly used Hoechst 33342 (also available from Biotium), which remains the better choice for nuclear staining of cells that will be cultured for more than a few hours. Like all far-red dyes, RedDotTM1 and RedDotTM2 fluorescence is not visible to the human eye, and must be imaged using a CCD camera or confocal microscope in the Cy5 channel. On the plus side, RedDotTM1 and RedDotTM2 emission spectra are very well separated from green fluorescent dyes and probes, further reducing the potential for artifacts arising from fluorescence cross-talk. In addition, they leave the visible blue fluorescence channel free for detection of other targets.
Learn more about Biotium's selection of novel nuclear stains for microscopy or flow cytometry.
Karg, T.J. and Golic, K.G. (2018). Photoconversion of DAPI and Hoechst dyes to green and red-emitting forms after exposure to UV excitation. Chromosoma 127 (2): 235245.
Roberts, L. (2019). Roberts et al. 2019 Microscopy Today, 27(5), 18-24. doi:10.1017/S1551929519000804
Żurek-Biesiadaurek, D., Kędracka-Krok, S. and Dobrucki, J. W. (2013). UV-activated conversion of Hoechst 33258, DAPI, and Vybrant DyeCycle fluorescent dyes into blue-excited, green-emitting protonated forms. Cytometry, 83A: 441451. doi:10.1002/cyto.a.22260
Download the RedDotTM1 and RedDotTM2 flyer
TOTO and TO-PRO are trademarks or registered trademarks of Thermo Fisher Scientific; Cy Dye is a registered trademark of Cytiva.