Content #1
Content #1
Content #1
Centrioles are cylindrical organelles composed primarily of tubulin. They are found in most eukaryotic cells, and bound pairs of centrioles comprise a centrosome structure. The proximal end of centrioles establishes a highly structured matrix called the pericentriolar material (PCM), which is the site of microtubule (MT) nucleation and centriole duplication. The distal end of centrioles assembles two different types of projections: distal and subdistal appendages (DAs and SDAs, respectively). DAs mediate the attachment of ciliary vesicles and their subsequent fusion with the cytoplasmic membrane, while SDAs anchor MTs and position centrioles and cilia. Centriole appendages thus play a necessary role in cellular processes such as development, motility, signaling, and maintenance of cellular architecture, making knowledge of their structure and dynamics applicable to several human health conditions. However, owing to their small size and biochemical complexity, appendage architecture and the mechanisms guiding centriole construction and maintenance remain mostly unknown.
In two striking reports published in JCB and Nature Communications, researchers from the Laboratory of Protein Dynamics and Signaling (NIH/NCI/CCR, Frederick, Maryland) shed new light on centriole assembly and composition using correlative advanced microscopy techniques. Antibody conjugates of Biotium’s CF®647 Dye played a crucial role in both of these studies, as it was the sole fluorophore used in STORM for all protein targets. In the first publication, the group used correlative super-resolution STORM and electron microscopy (EM) to accurately determine appendage protein localization in relation to centriole microtubules. Along with illustrating the early assembly of SDAs via recruitment of the protein Odf2 to MTs, followed by recruitment of the centriole-constructing proteins CCDC68, CCDC120, Cep170, Cep128, and Ninein (Figure 1), they discover a novel distal appendage protein ANKRD26 and precisely detail its involvement during early distal appendage assembly. Furthermore, they show that the appendage undergoes dramatic reorganization before mitosis and that mammalian appendages associate with centriole microtubule triplets via a filamentous base. In a subsequent publication, members of the same group used STORM imaging coupled with cells genetically engineered for the rapid degradation of the protein CPAP to show that CPAP is critical for the assembly of human centrioles and answer why CPAP mutations are associated with human diseases. Together, these studies utilize advanced high-resolution microscopy techniques to challenge the traditional understanding of mammalian distal appendage assembly and maintenance throughout mitosis. The studies also provide insight into how abnormalities in this process can impact human health.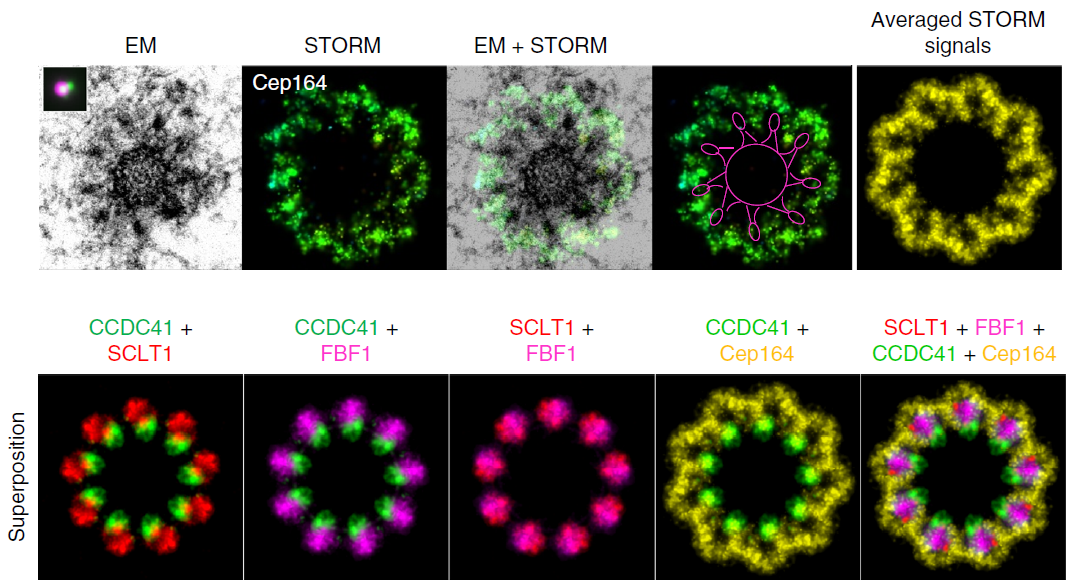 Fig. 3 Correlative STORM/EM analysis of centriole appendage assembly. RPE-1 C1-GFP cells were immunolabeled for the indicated proteins and imaged using 3D STORM and correlative electron microscopy analysis. The top panel shows one 80 nm section containing DA EM densities, the STORM image of the same centriole (stained for Cep164), the merged STORM and EM image, the STORM image with a line delineating the centriole and the DA head densities, and the averaged STORM signal. In the bottom panel, averaged STORM images for each protein target were superimposed to generate a horizontal distribution map of their localization patterns. All STORM images come from data collected using Biotium’s CF®647 F(ab’)2 secondary antibodies along with the appropriate primary antibodies and were pseudo-colored for display purposes. For scale, each image shown represents approximately 650 nm across at the bottom. Credit: Modified from Bowler et al. https://doi.org/10.1038/s41467-018-08216-4 reproduced under the Creative Commons license.
Fig. 3 Correlative STORM/EM analysis of centriole appendage assembly. RPE-1 C1-GFP cells were immunolabeled for the indicated proteins and imaged using 3D STORM and correlative electron microscopy analysis. The top panel shows one 80 nm section containing DA EM densities, the STORM image of the same centriole (stained for Cep164), the merged STORM and EM image, the STORM image with a line delineating the centriole and the DA head densities, and the averaged STORM signal. In the bottom panel, averaged STORM images for each protein target were superimposed to generate a horizontal distribution map of their localization patterns. All STORM images come from data collected using Biotium’s CF®647 F(ab’)2 secondary antibodies along with the appropriate primary antibodies and were pseudo-colored for display purposes. For scale, each image shown represents approximately 650 nm across at the bottom. Credit: Modified from Bowler et al. https://doi.org/10.1038/s41467-018-08216-4 reproduced under the Creative Commons license.
Related Technologies
Learn more about Biotium’s high-performance CF® Dyes with multi-color flexibility, CF® Dye-conjugated secondary antibodies, and reagents for immunofluorescence microscopy. We also offer several product lines that have been validated for STORM and other super-resolution microscopy techniques.
Full Citations
Bowler, M., Kong, D., Sun, S., Nanjundappa, R., Evans, L., Farmer, V., … & Loncarek, J. (2019). High-resolution characterization of centriole distal appendage morphology and dynamics by correlative STORM and electron microscopy. Nature communications, 10(1), 993. https://doi.org/10.1038/s41467-018-08216-4
Vásquez-Limeta, A., Lukasik, K., Kong, D., Sullenberger, C., Luvsanjav, D., Sahabandu, N., … & Loncarek, J. (2022). CPAP insufficiency leads to incomplete centrioles that duplicate but fragment. Journal of Cell Biology, 221(5), e202108018. https://doi.org/10.1083/jcb.202108018