Content #1
Content #1
Content #1
It is known that cells can assemble macromolecular complexes into scaffoldings that serve as substrates for catalytic processes. While ample molecular neurobiology research indicates that neurotransmission depends on scaffold-based optimization strategies, the precise molecular landscape of the presynaptic active zone (AZ) where neurotransmitter is released upon synaptic vesicle (SV) fusion has yet to be visualized. MINFLUX (Minimal photon FLUX) is a super-resolution microscopy method that combines aspects of single-molecule localization microscopy (SMLM) and Stimulated Emission Depletion (STED) microscopy to achieve higher spatial and temporal resolution, while also requiring fewer fluorescently emitted photons. MINFLUX works best with dyes that meet the parameters for STORM imaging. In the last five years, MINFLUX has been used to uncover the spatiotemporal details of biological processes such as the bacterial type 3 secretion system (Carsten et al., 2022) and the “walking” of kinesin-1 down to the molecular level (Wolff et al., 2023).
Grabner et al. recently applied MINFLUX to reveal the long-unknown molecular details of the photoreceptor AZs in an article published in Science Advances. Following a novel sample immobilization technique for transferring layers of rod synaptic terminals onto glass coverslips from fresh slices of mouse retina, rod AZs were immunolabeled using antibodies against bassoon and RIM2 labeled with Biotium’s CF®660C and another fluorophore respectively and imaged by two-color 3D-MINFLUX. Using this technique, the team was able to achieve AZ labeling and imaging to nanometer-level 3D resolution (see Grabner Figure 7). This two-color imaging showed conclusively that the proteins bassoon and RIM2 colocalize in the AZ. Together with results from single-labeling experiments, these 3D-MINFLUX results showed that a molecular complex formed by the longitudinal repeats of the proteins bassoon–RIM2–ubMunc13-2–Cav1.4 forms the SV release site in rods. The authors then explained that their findings are in line with several other recent super-resolution studies of synapses from other organs/organisms and proposed that MINFLUX would be useful in further determining the molecular topography of AZs in these other systems. This adds to a growing number of reports that Biotium CF® Dyes excel in a variety of super-resolution applications, including STORM and STED (Figure 1).
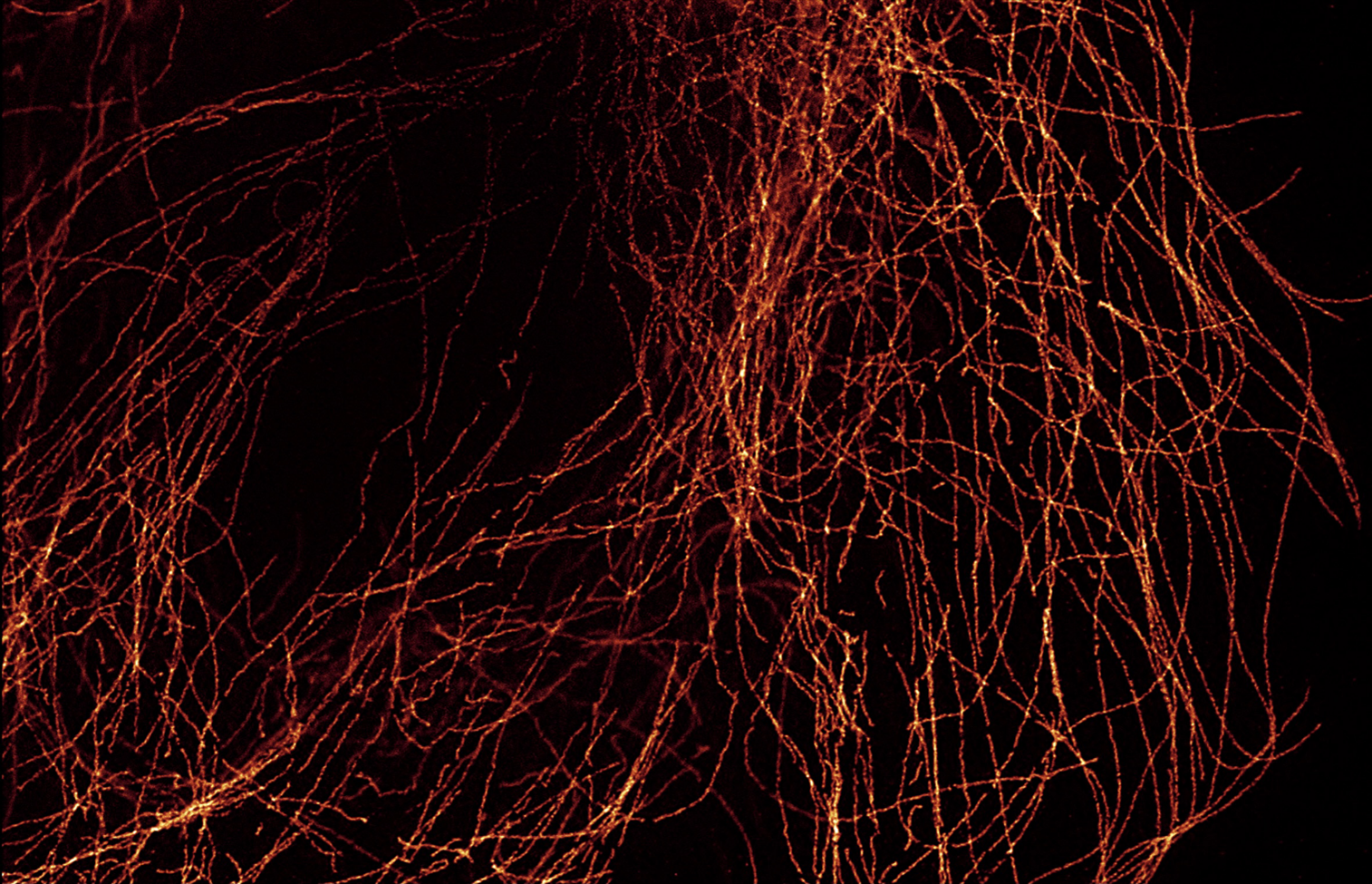
Learn more about Biotium’s high-performance CF® Dyes with multi-color flexibility. Certain CF® Dyes have been validated for super-resolution microscopy applications including STORM and STED, and we now offer Mix-n-Stain™ STORM CF® Dye Antibody Labeling Kits as part of our popular Mix-n-Stain™ Antibody Labeling Kit product line. We also sell a wide variety of other cellular probes validated for super-resolution microscopy for a variety of organelles and structures.
Full Citation
Grabner, C. P., Jansen, I., Neef, J., Weihs, T., Schmidt, R., Riedel, D., … & Moser, T. (2022). Resolving the molecular architecture of the photoreceptor active zone with 3D-MINFLUX. Science Advances, 8(28), eabl7560. DOI: 10.1126/sciadv.abl7560
Other References
Carsten, A., Rudolph, M., Weihs, T., Schmidt, R., Jansen, I., Wurm, C. A., … & Aepfelbacher, M. (2022). MINFLUX imaging of a bacterial molecular machine at nanometer resolution. Methods and Applications in Fluorescence, 11(1), 015004. https://doi.org/10.1088/2050-6120/aca880
Wolff, J. O., Scheiderer, L., Engelhardt, T., Engelhardt, J., Matthias, J., & Hell, S. W. (2023). MINFLUX dissects the unimpeded walking of kinesin-1. Science, 379(6636), 1004-1010. https://doi.org/10.1126/science.ade2650