The imaging resolution of traditional optical microscopy is limited by the diffraction limit of light. Recent developments in super-resolution microscopy have led to new techniques which bypass the diffraction limit problem and increase the resolving power of optical microscopes. One such technique, stochastic optical reconstruction microscopy (STORM), has permitted the super-resolution imaging of small cellular structures in staggering multi-color detail. However, imaging cells and tissues with STORM is limited to a shallow depth of ~1 μm, and thus thin sectioning and stitching axial stacks must be used to obtain super-resolution images deep into these samples.
In a recent study, Kim and colleagues from the University of California at Berkeley have combined STORM and wide-field oblique plane microscopy (OPM) to create oblique-plane STORM (obSTORM). This novel super-resolution microscopy technique uses oblique light-sheet illumination and oblique fluorescence detection to image deep into thick biological samples without extensive sample preparation or axial stacking. In the paper, secondary antibodies conjugated to CF®568 were used to label microtubules or mitochondria in A549 cells on coverslips. Using obSTORM, the researchers demonstrated super-resolution imaging at a depth of 32 μm, more than an entire A549 cell thickness. The obSTORM technology enables facile cross-sectional super-resolution imaging through thicker cells and tissue samples on easily prepared coverslips.
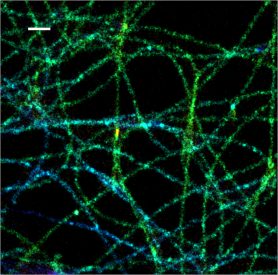
Full citation
J. Kim, M. Wojcik, Y. Wang, K. Xu and X. Zhang, “Oblique plane single-molecule localization microscopy for thick samples,” bioRxiv (https://doi.org/10.1101/289686)

