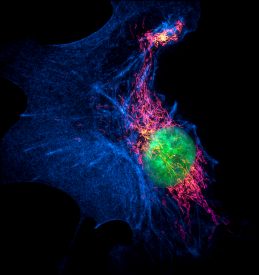
A deeper understanding of cell function requires determination of molecular-scale architecture. This includes interactions of subcellular organelles and biomolecular protein assemblies. While fluorescence super-resolution imaging techniques have significantly improved upon the resolution limit of conventional light microscopy, they still lack the resolution achievable by electron microscopy (EM). However, EM lacks the tools that provide molecular specificity provided by fluorescence imaging techniques.
In a recent Nature Methods publication, Zhang et al. introduce a super-resolution (SR) microscope that enables high-quality 3D-multicolor fluorescence imaging at nanoscale resolution. Their invention relies on two previously developed SR techniques, interferometric 4Pi single molecule switching (SMS) in combination with refined ratiometric detection that takes advantage of ‘salvaged fluorescence’ (SF). SMS relies on imaging spectrally similar, single laser excitable fluorophores simultaneously. 4Pi-SMS improves axial localization precision. Ratiometric SF recovers previously lost reflected fluorescence, allowing spectral identification of fluorophores from on their emission profiles. Many of the best-performing dyes for SMS are clustered in the far-red range.The authors use Alexa Fluor® 647 (AF647), CF®660C, Dyomics 634 (DY634), Dylight® 650 (DL650) and CF®680 for their 4Pi-SMS SF approach. All dyes were efficiently and simultaneously excited at 642 nm and showed minimal cross-talk, for 2- or 3-color imaging of various cellular substructures. Dual color (AF647 and CF®660C) or triple color (DY634, DL650 and CF®680) immunolabeling was used to resolve ER tubular membranes and microtubules. They demonstrate the power of this technique to resolve several spacially close structures in ER membranes, mitochondria, and a synaptonemal complex. Furthermore, the authors applied the technique toward imaging the Golgi apparatus and ER-plasma membrane contact sites, a particularly challenging structure for imaging that was previously only possible through EM. The authors also address the advantages of CF®660C dye for ratiometric imaging with SF, and the use of nanobodies, genetic tags (HALO® or SNAP®), or click-chemistry in place of primary and secondary antibody labeling to reduce displacement in target localization.
This technique provides an invaluable tool for the study of cellular structures at a level of detail previously impossible. Their SF approach can be adapted with minor optical modifications toward implementation in single objective systems and would be amenable to live cell imaging using photoactivable fluorescent dyes.
Learn more about our CF® dye options for super-resolution microscopy, CF® dye technology and CF® dye antibody and other conjugates for fluorescence imaging.