Single-molecule tracking (SMT) is an effective tool for probing molecular dynamics in cells. SMT becomes especially powerful if multiple molecular species can be analyzed simultaneously, enabling more complex, multi-component interactions to be studied in intact biological systems. Multicolor SMT, however, remains challenging: spectral cross talk between multiple fluorophores limits scalability and the need for multiple lasers for excitation complicates the optical setup.
A recent paper describes a multicolor spectral imaging system, compatible with a commercial inverted microscope, for single-molecule SMT in live cells. Using a single 637 nm laser, the authors were able to simultaneously excite and image a panel of spectrally proximal fluorophore-tagged probes, CF®633-WGA, CellMask DR, and CF®680-Transferrin, which emit at wavelengths only 15–30 nm apart. Spatial and spectral resolution were comparable to that obtained with STORM, with minimal spectral crosstalk. WGA and transferrin appeared to be almost completely immobile, suggesting localization to nano-domains such as ‘lipid rafts’ or interaction with cellular scaffolds.
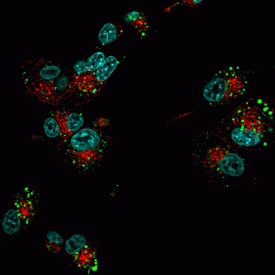
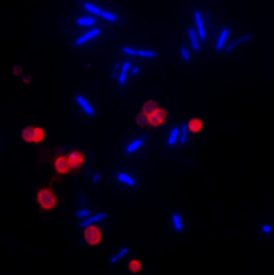
Left: U87-MG cells stained with CF®680R transferrin (red), CF®488A dextran (green), and Hoechst (blue). Right: E. coli and Staphylococcus stained with CF®633-WGA (red) and DAPI (blue).
To read the original article, click here.
Huang T et al. Simultaneous Multicolor Single-Molecule Tracking with Single-Laser Excitation via Spectral Imaging. Biophys J. 2018;114(2):301-310. doi: 10.1016/j.bpj.2017.11.013.
Learn more about Biotium’s CF® dye labeled bioconjugates and CF® dyes for super-resolution microscopy.

