ViaFluor® SE Cell Proliferation Kits
Fixable cytoplasmic stains for monitoring cell division by flow cytometry. The dyes can also be used to track cell populations in co-culture.
Please fill in the inquiry form and we will contact you shortly.
Wishlist updated! View wishlist
Product Description
ViaFluor® SE Cell Proliferation Kits use amine-reactive dyes to covalently label cells throughout the cell cytoplasm and intracellular compartments for fixable fluorescent staining. Cell proliferation dyes are commonly used to monitor cell division by flow cytometry. The dyes also can be used to stably label cells to image cell morphology, or to track cell populations in mixed co-culture experiments.
Features
- Non-toxic dyes covalently label cell cytoplasm for fixable staining
- Track cell proliferation in vivo or in vitro by dye dilution using flow cytometry
- Long term imaging of cell morphology or co-cultures by microscopy
- Excellent performance & lower cost compared to leading competitors
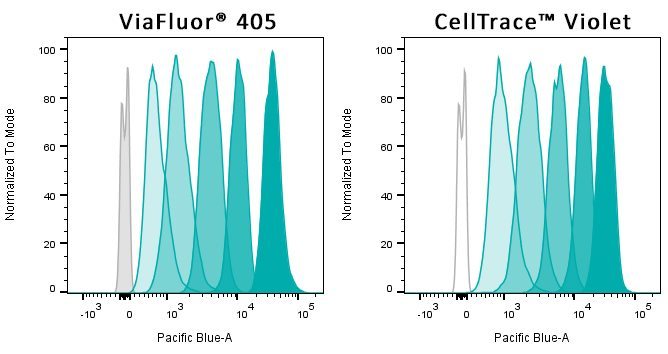
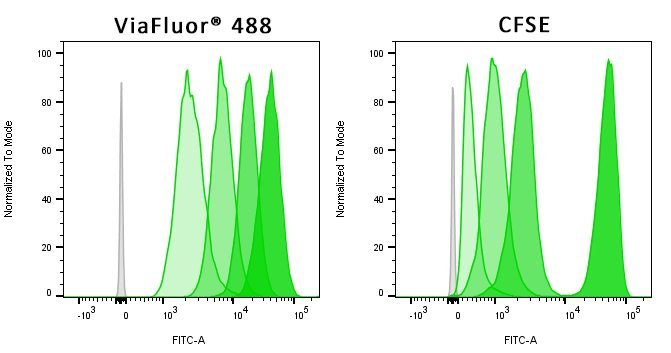
- ViaFluor® 405 & ViaFluor® 488 SE are much less toxic than CFSE
Kit Components
- Lyophilized dye in single use vials
- Anhydrous DMSO for dissolving dye
Spectral Properties (Ex/Em after hydrolysis)
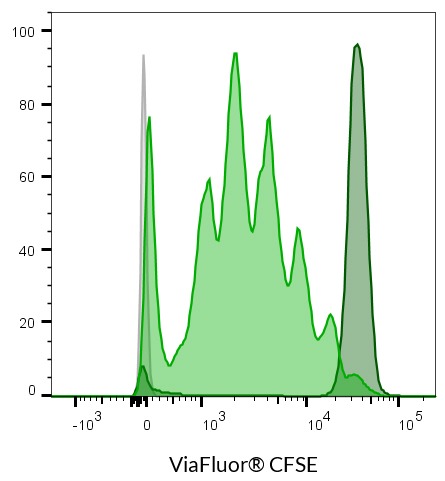
- ViaFluor® CFSE: 495/515 nm
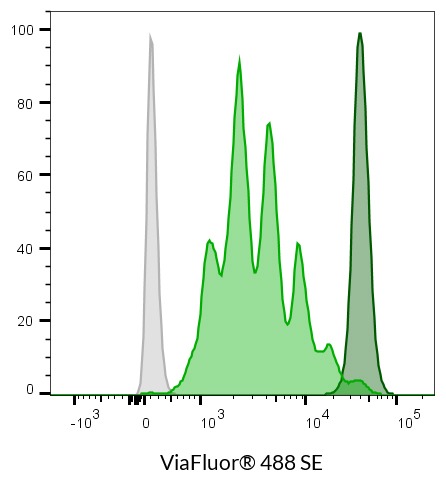
- ViaFluor® 488 SE: 493/532 nm
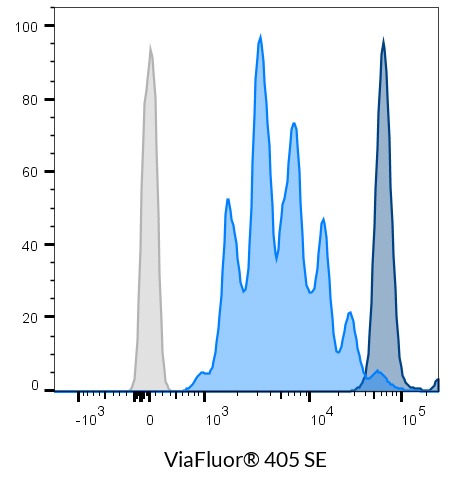
- ViaFluor® 405 SE: 408/452 nm
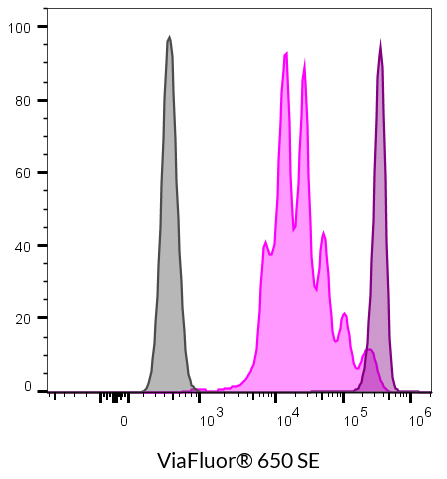
- ViaFluor® 650 SE: 653/682
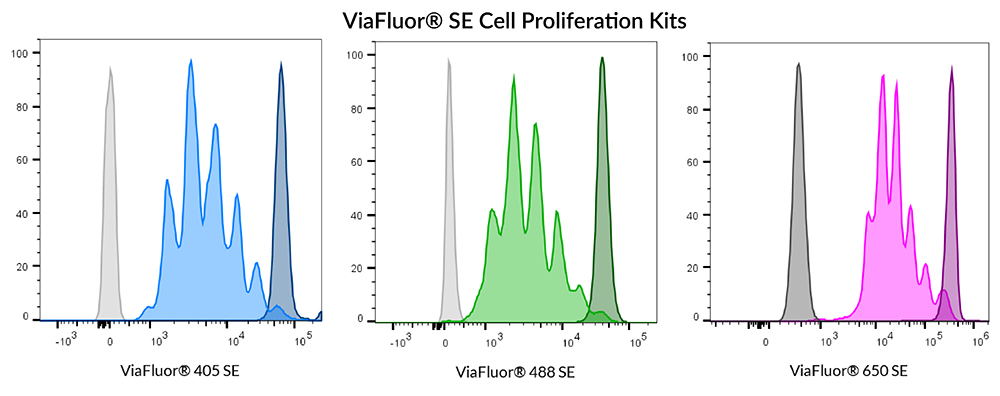
ViaFluor® SE Dyes for Cell Proliferation Tracking
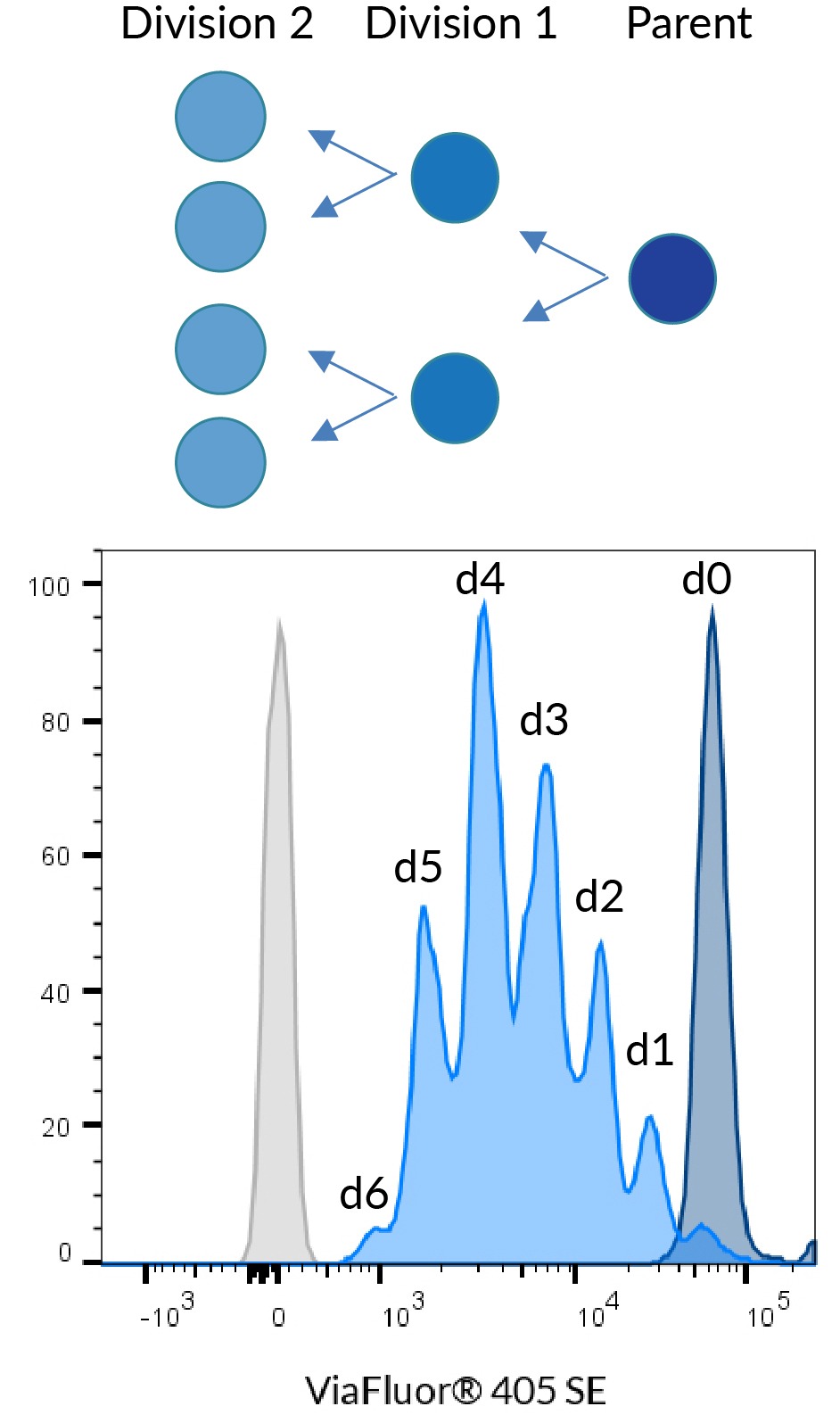
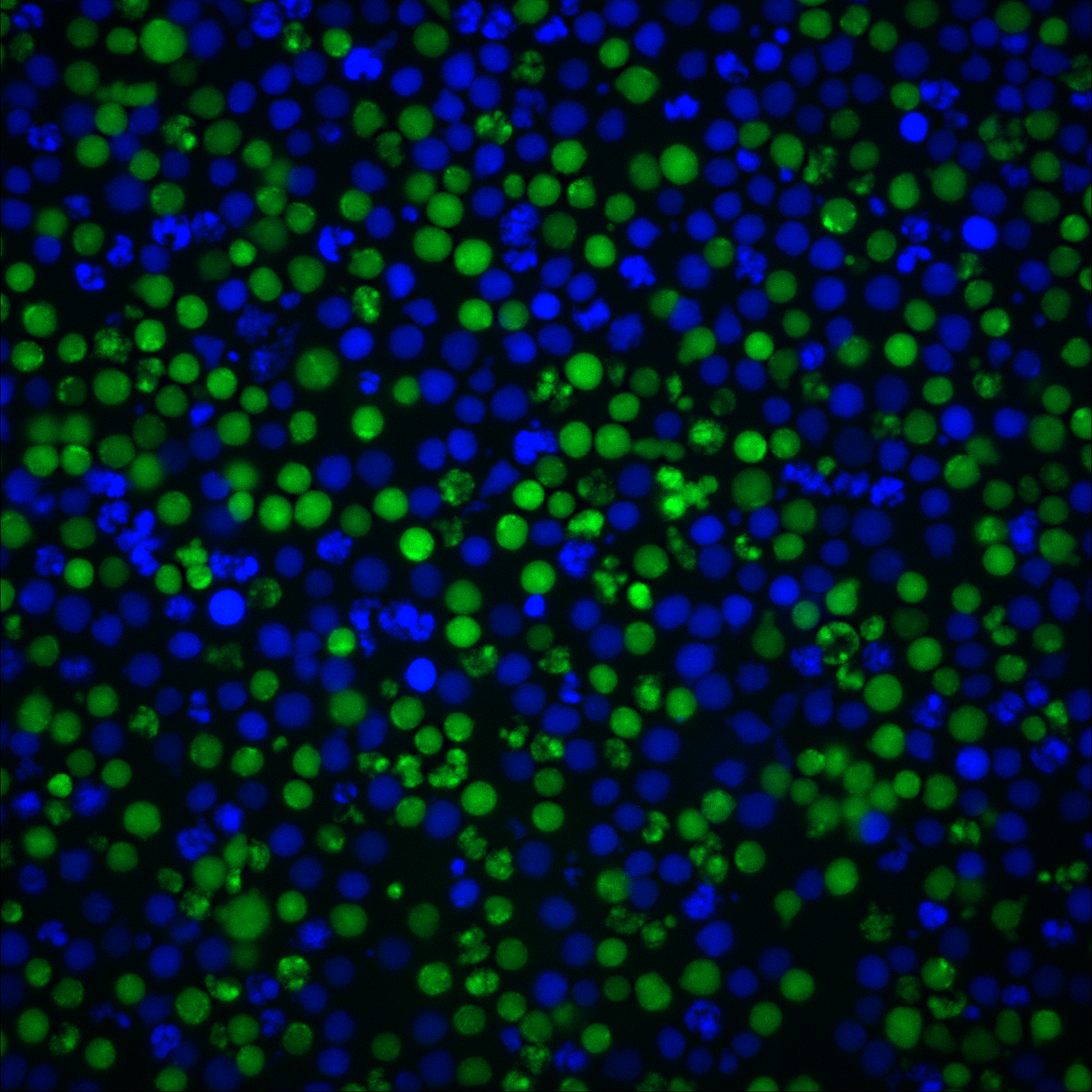
ViaFluor® SE dyes are membrane-permeant compounds that are initially non-fluorescent esters, but are converted to fluorescent dyes by intracellular esterases and will covalently react with amine groups on intracellular proteins at the same time, forming fluorescent conjugates that are retained in the cell. Immediately after staining, a single bright fluorescent population will be detected by flow cytometry. With each cell division, daughter cells inherit roughly half of the fluorescent label, allowing the number of cell divisions that occur after labeling to be detected by the appearance of successively dimmer fluorescent peaks on a flow cytometry histogram compared to cells analyzed immediately after staining. Thus, cell proliferation dyes can be used to track multiple cell divisions of cells grown in culture or injected in vivo after labeling with the ViaFluor® SE dye.
The number of assays that can be performed per kit depends on the dye concentration used (see the product protocol for more information). When used at 1 uM to label 106 cells in one mL, each dye vial can be used for 90-100 labelings.
Other Applications for ViaFluor® SE Dyes
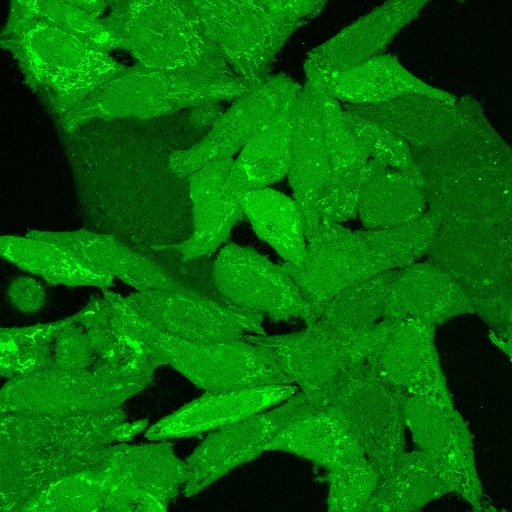
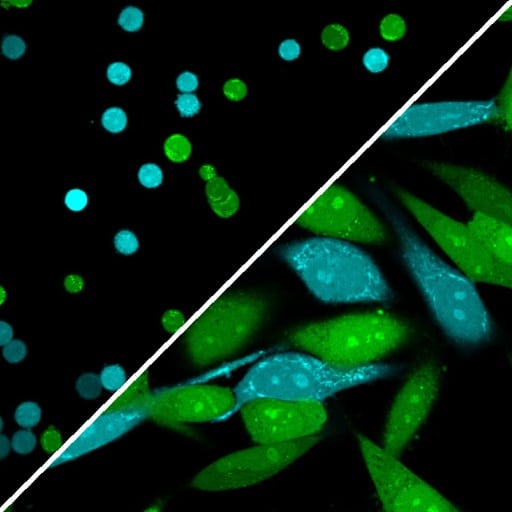
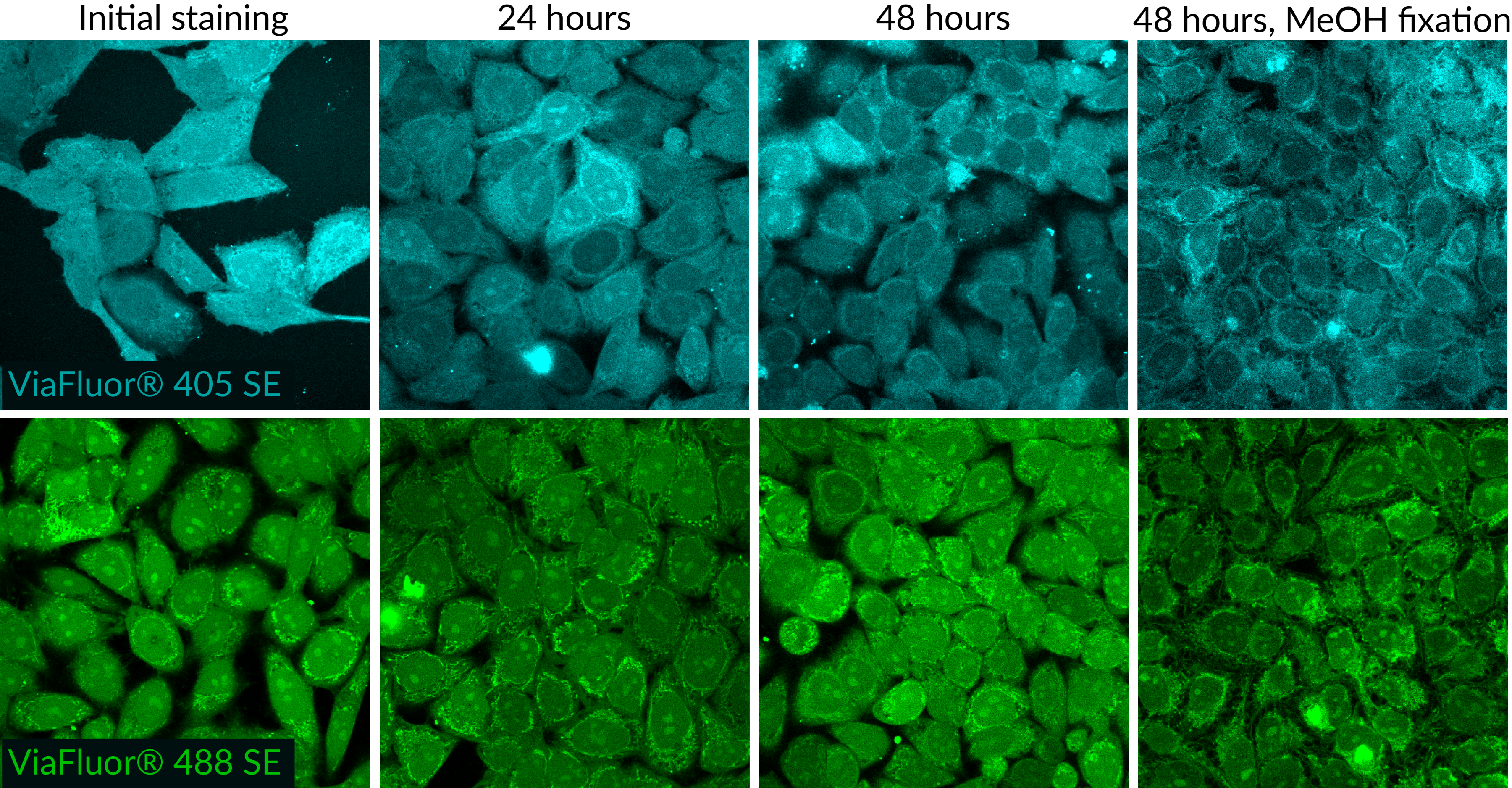
ViaFluor® SE dyes also can be used for imaging cell morphology or identifying cells in co-culture by microscopy. Because the staining is non-toxic and well-retained, it can be used for imaging live cells over time. See our Tech Tip: Using ViaFluor® SE Stains for Cell Tracing and Co-Culture.
All three ViaFluor® SE dyes can stain gram-positive bacteria, but not gram-negative bacteria. ViaFluor® CFSE stains the cytoplasm in yeast, but ViaFluor® 405 & ViaFluor® 488 stain the yeast cell periphery. See our Cellular Stains Table for more information on how our dyes stain various organisms.
Cell Division | Catalog No. | Ex/Em (nm) | Flow detection | Features |
|---|---|---|---|---|
| ViaFluor® 405 Cell Proliferation Kit | 30068 | 408/452 | Pacific Blue® | • Track cell division by dye dilution using flow cytometry • ViaFluor® 488 is a unique, improved green dye to replace CFSE • ViaFluor® 405 replaces CellTrace™ Violet • ViaFluor® 650 replaces CellTrace™ Far Red |
| ViaFluor® 488 Cell Proliferation Kit | 30086 | 493/532 | FITC | |
| ViaFluor® 650 Cell Proliferation Kit | 30139 | 653/682 | APC | |
| ViaFluor® CFSE Cell Proliferation Kit | 30050 | 495/519 | FITC |