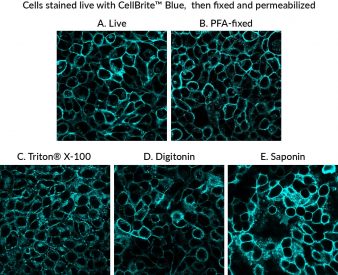
CellBrite® Cytoplasmic Membrane Dyes are membrane dyes that are useful for visualizing cell outlines. However, because they are lipophilic, the dyes tend to redistribute after detergent permeabilization (Fig. 1). For membrane staining of live cells followed by fixation and immunofluorescence, we recommend our newer CellBrite® Fix and MemBrite® Fix fixable cell surface stains. However, these stains cannot be used on cells after they are already fixed. In contrast, our original CellBrite® Cytoplasmic Membrane Dyes can be used to stain cells after formaldehyde fixation. However, their poor tolerance for detergent can make it challenging to incorporate them into an intracellular immunofluorescence protocol.
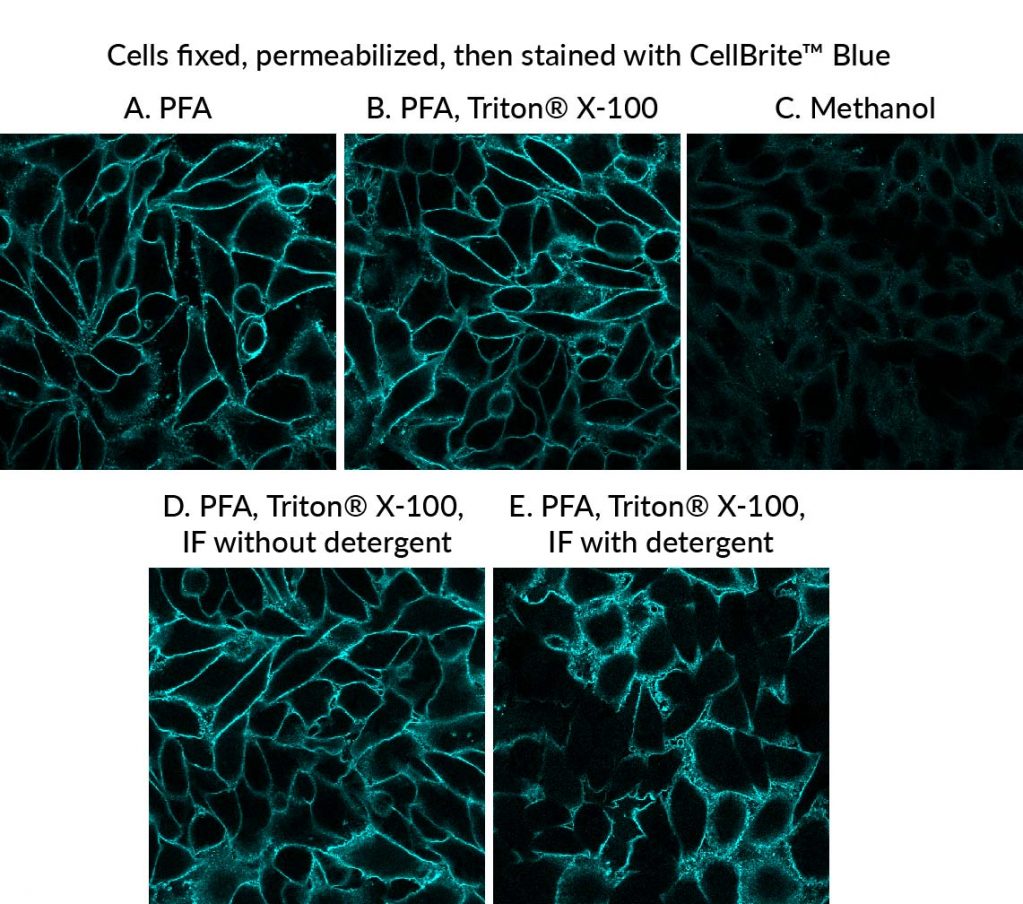
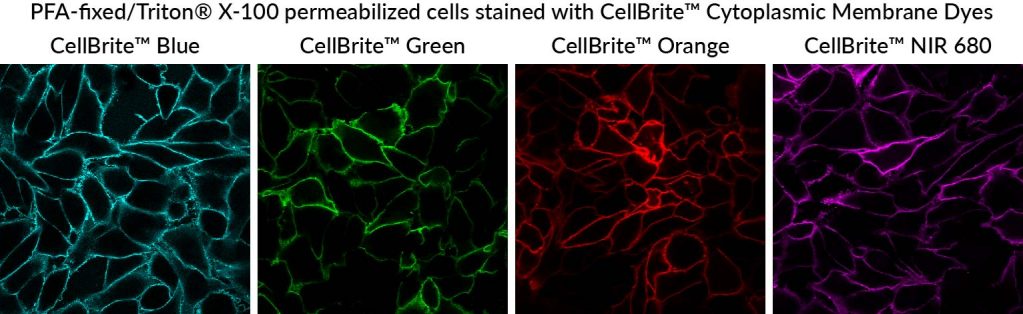
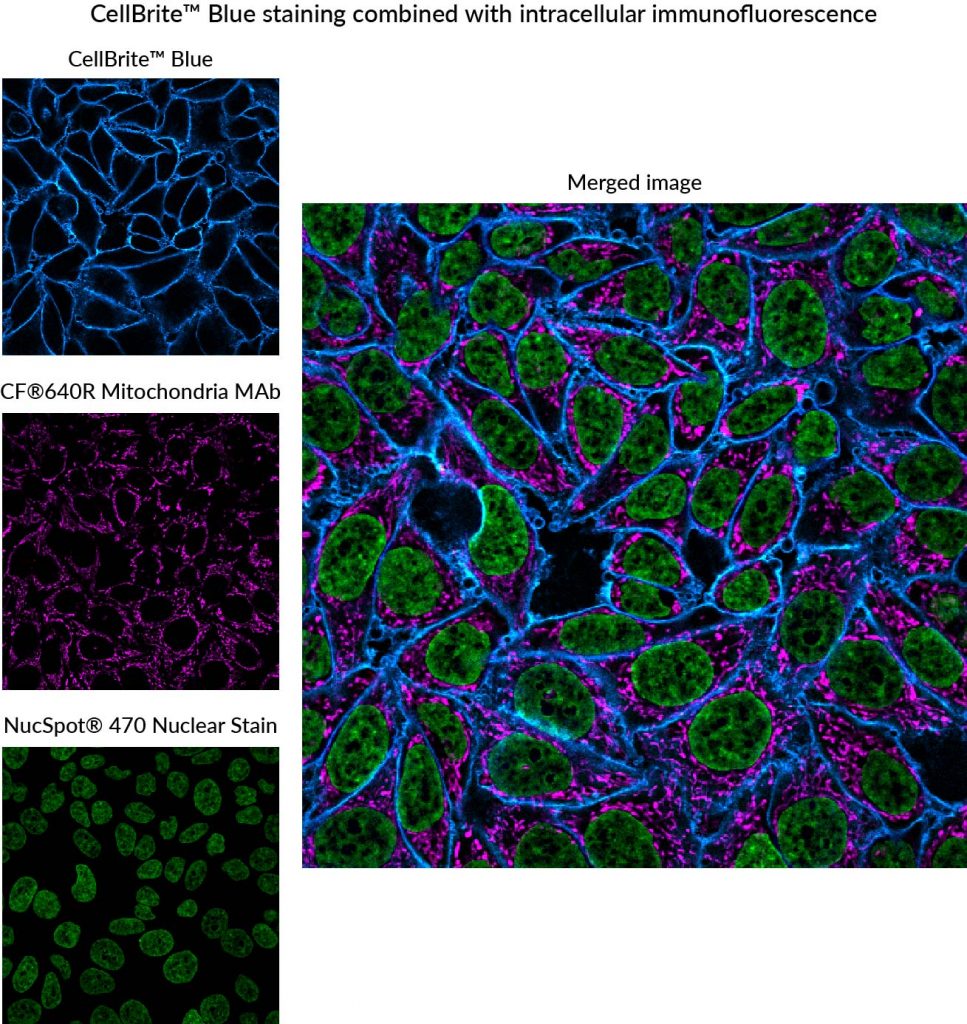
We have developed the following protocol to fix and permeabilize cells before staining with CellBrite® Cytoplasmic Membrane Dyes. Even though the cells are permeabilized before the membrane dye is applied, staining still primarily localizes to the plasma membrane, allowing visualization of cell boundaries (Fig. 2). This protocol works well with a variety of CellBrite® Dyes (Fig. 3), and the staining is well-retained during a typical antibody staining protocol (Figs. 2, 4). However, to avoid dye redistribution, it is important to avoid using detergents after the CellBrite® staining step (see Fig. 2, D-E).
Protocol:
- Rinse live cells 3X with HBSS buffer.
- Fix cells with 4% paraformaldehyde in PBS, 20 minutes at room temperature.
Note: You may use your preferred formaldehyde fixation protocol, but alcohol or solvent fixatives are not recommended (see Figure 2C). - Rinse cells 3X with PBS.
- Permeabilize cells with 0.1% Triton X-100 in PBS for 10 minutes at room temperature.
Note: Permeabilization may be performed at 4°C if preferred. - Rinse cells 3X with PBS.
- Prepare CellBrite® dye staining solution in PBS according to the product protocol.
Download a protocol:
CellBrite® Green, Orange, or Red Protocol
CellBrite® Blue Protocol
CellBrite® NIR dyes Protocol - Add CellBrite® staining solution to the cells and incubate in the dark for 10 minutes at room temperature.
- Rinse cells 3X with PBS.
- Proceed to blocking, antibody incubation, and washing according to your usual immunofluorescence protocol.
Important: Do not include detergent in any of the buffers used after the CellBrite® staining step.
Note: Protect samples from light. - Image cells in PBS. Glycerol-based mounting medium is not recommended for use with CellBrite® dyes.