Cytochrome c Monoclonal Mouse Antibody (CYCS/1010)
Wishlist updated! View wishlist
Product Description
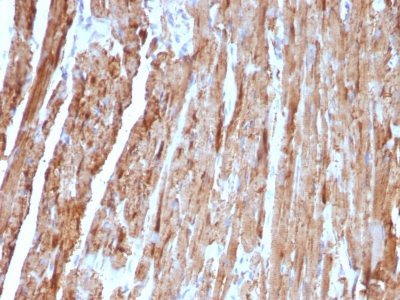
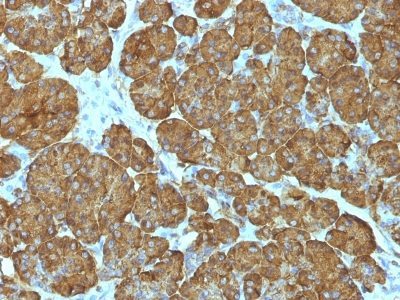
Cytochrome c is a well-characterized mobile electron transport protein that is essential to energy conversion in all aerobic organisms. In mammalian cells, this highly conserved protein is normally localized to the mitochondrial inter-membrane space. More recent studies have identified cytosolic cytochrome c as a factor necessary for activation of apoptosis. During apoptosis, cytochrome c is trans-located from the mitochondrial membrane to the cytosol, where it is required for activation of caspase-3 (CPP32). Overexpression of Bcl-2 has been shown to prevent the translocation of cytochrome c, thereby blocking the apoptotic process. Overexpression of Bax has been shown to induce the release of cytochrome c and to induce cell death. The release of cytochrome c from the mitochondria is thought to trigger an apoptotic cascade, whereby Apaf-1 binds to Apaf-3 (caspase-9) in a cytochrome c-dependent manner, leading to caspase-9 cleavage of caspase-3. This MAb recognizes total cytochrome C which includes both apocytochrome (i.e. cytochrome in the cytosol without heme attached) and holocytochrome (i.e cytochrome in the mitochondria with heme attached).
Primary antibodies are available purified, or with a selection of fluorescent CF® dyes and other labels. CF® dyes offer exceptional brightness and photostability. See the CF® Dye Brochure for more information. Note: Conjugates of blue fluorescent dyes like CF®405S and CF®405M are not recommended for detecting low abundance targets, because blue dyes have lower fluorescence and can give higher non-specific background than other dye colors.
Catalog number key for antibody number 1010, Anti-Cytochrome c (CYCS/1010)
Catalog Number Key for Primary Antibody Conjugates
| Antibody # prefix | Conjugation | Ex/Em (nm) | Laser line | Detection channel | Dye Features |
|---|---|---|---|---|---|
| BNC04 | CF®405S | 404/431 | 405 | DAPI (microscopy), AF405 | CF®405S Features |
| BNC88 | CF®488A | 490/515 | 488 | GFP, FITC | CF®488A Features |
| BNC68 | CF®568 | 562/583 | 532, 561 | RFP, TRITC | CF®568 Features |
| BNC94 | CF®594 | 593/614 | 561 | Texas Red® | CF®594 Features |
| BNC40 | CF®640R | 642/662 | 633-640 | Cy®5 | CF®640R Features |
| BNC47 | CF®647 | 650/665 | 633-640 | Cy®5 | CF®647 Features |
| BNCB | Biotin | N/A | N/A | N/A | |
| BNUB | Purified | N/A | N/A | N/A | |
| BNUM | Purified, BSA-free | N/A | N/A | N/A |