Lectin conjugates, such as WGA (wheat germ agglutinin) or Con A (concanavalin A), bind with a high specificity to sugar groups on glycoproteins. WGA lectins bind to sialic acid and N-acetylglucosamine residues, while Con A lectins bind to a-mannopyranosyl and a-glucopyranosyl residues. Due to the expression of glycoproteins on cell membranes, conjugated lectins are widely used in fluorescence microscopy for staining cellular surfaces or intracellular organelles. Unlike lipophilic dyes such as DiI, DiO, or CellBrite® Cytoplasmic Membrane Dyes, an advantage of using lectin conjugates is their tolerance for permeabilization – allowing co-staining with antibodies for immunofluorescence. In this article, we provide considerations and a general protocol for staining cellular surfaces with lectin conjugates.
Summary:
- WGA and Con A conjugates label cell surface glycoproteins in live or fixed cells, making them versatile probes for combining with antibody staining.
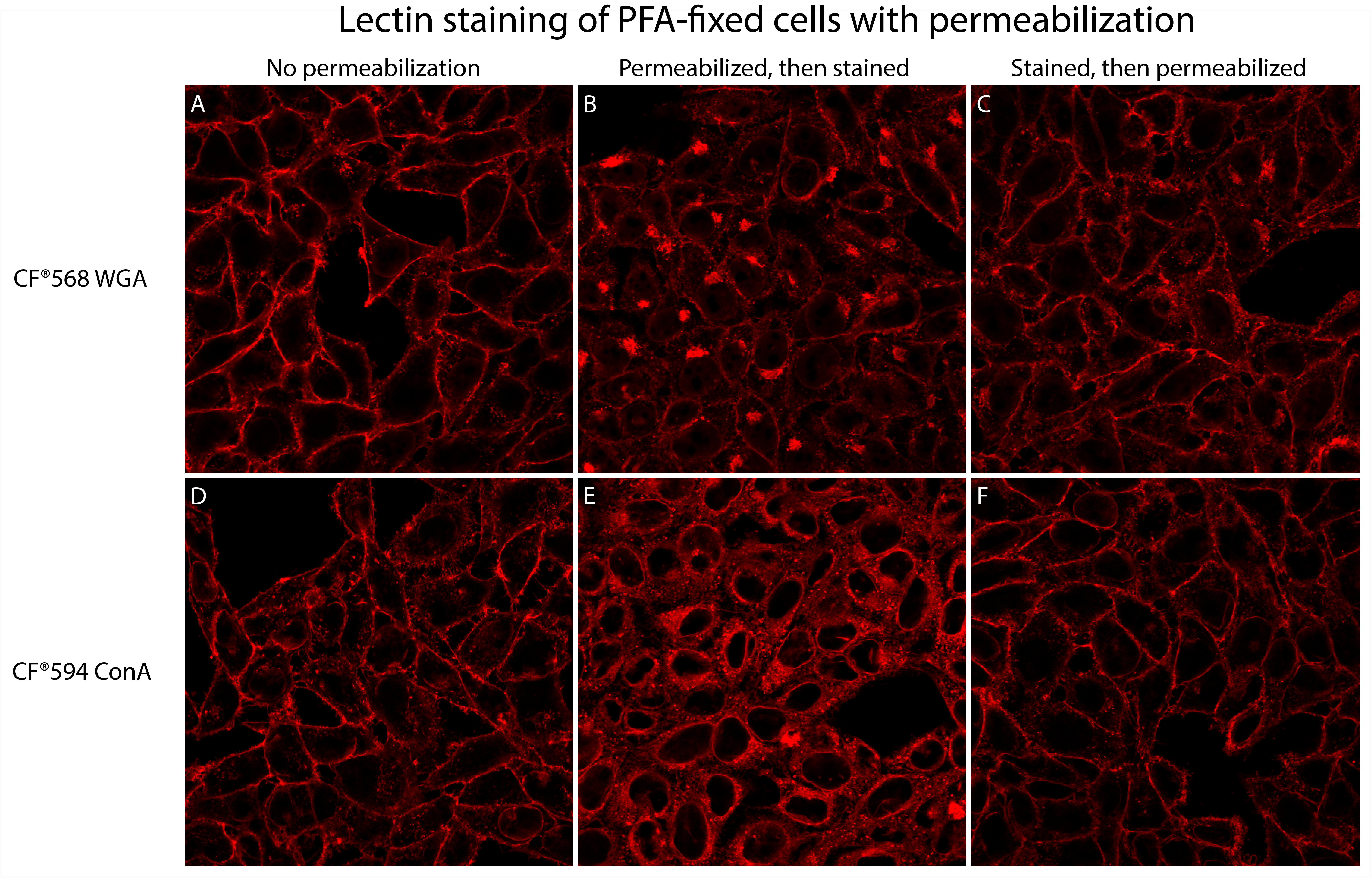
- In live cells, lectins preferentially stain the cell surface, but in fixed and permeabilized cells, they also stain intracellular organelles in the secretory pathway.
- Permeabilizing fixed cells after staining with lectins, rather than before, can help preserve selective plasma membrane staining (see Fig. 1).
- Cell staining with lectins will vary by cell type, particularly for primary cell lines or tissue sections. We recommend consulting the literature for information about lectin binding to your cells of interest before choosing a stain.
Procedure:
- Remove cell culture medium from live cells and rinse cells 3X with HBSS buffer.
- Fix cells with 4% paraformaldehyde in PBS for 20 minutes at room temperature.
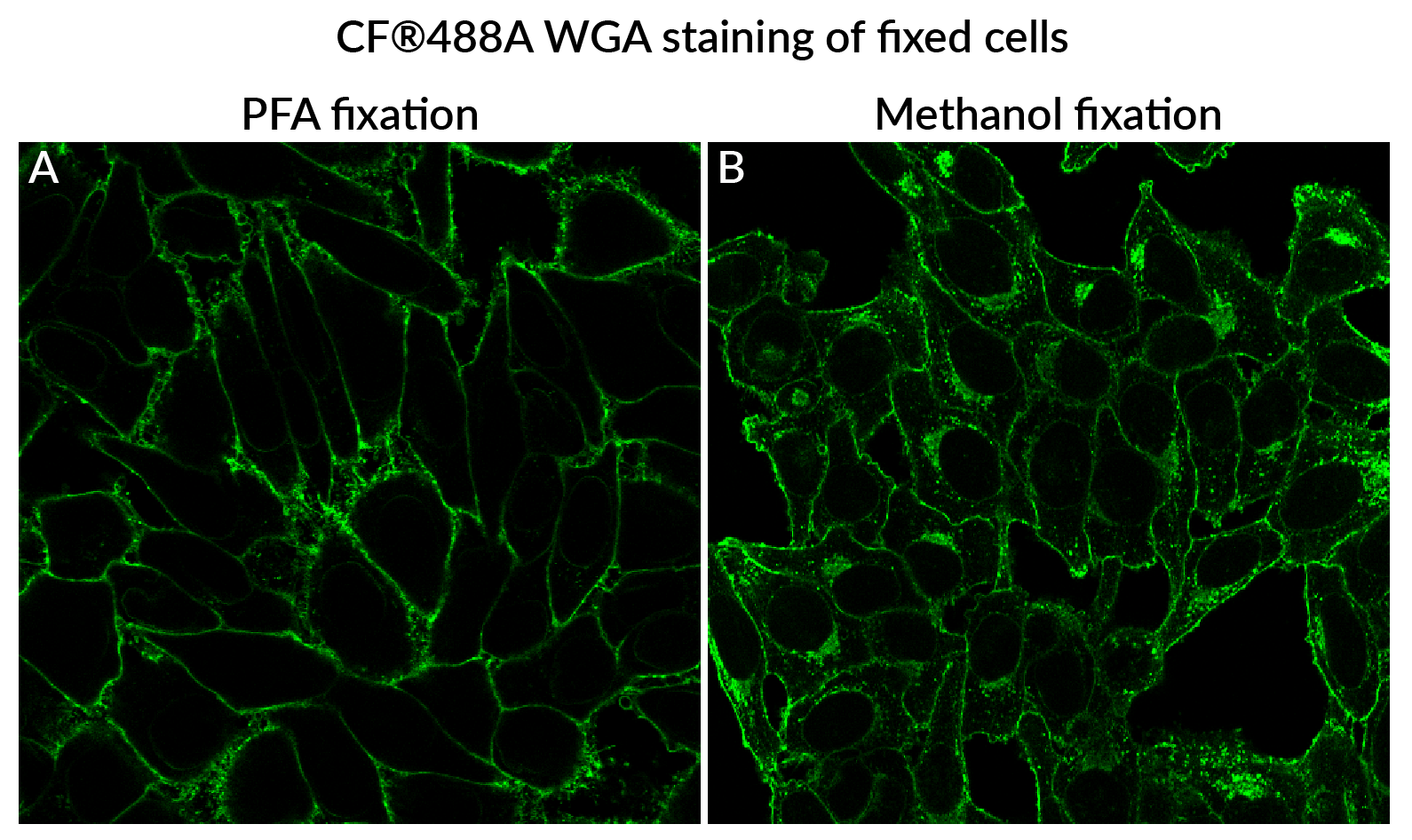
Note: When possible, use paraformaldehyde (PFA) fixation. Using alcohol or solvent fixatives (like methanol or acetone) will permeabilize the cells, and result in more intracellular staining compared to PFA (see Fig. 2).
Note: Avoid over-fixation with paraformaldehyde, which can increase permeability of the plasma membrane and may result in increased intracellular staining. - Rinse cells 3X with PBS.
- Stain cells with CF® Dye WGA or CF® Dye Con A in PBS for 10-30 minutes at room temperature.
Recommended concentrations: 1-5 ug/mL for WGA, or 50-200 ug/mL for Con A. - Rinse cells 3X with PBS.
- Permeabilize cells with 0.1% Triton X-100 in PBS (or use Biotium’s Permeabilization Buffer) for 30 minutes at room temperature.
- Rinse cells 3X with PBS.
- Proceed with immunofluorescence staining.
Note: We recommend omitting detergent from subsequent blocking and washing steps if possible, to minimize potential redistribution or loss of lectin signal.