Content #1
Content #1
Content #1
There are reportedly more than a billion tissue samples archived in hospitals and tissue banks around the world, representing a wealth of rich clinical data.1 Most of these samples are formalin-fixed and paraffin-embedded (FFPE). This preservation technique, developed over 100 years ago, remains the gold standard for diagnostics in pathology. FFPE tissue samples are regularly used to diagnose and study disease processes in clinical settings and FFPE processing is common practice for tissue preservation in many non-clinical research laboratories.2 FFPE processing is advantageous for many reasons: it allows for the preservation of morphology, provides convenient long-term storage and sectioning, and reduces the risks associated with infectious agents.

Lab equipment for preparing biopsies for pathologists to read by scientists. Selective focus. trairut noppakaew/Shutterstock.com
With the modern rise of next-generation sequencing (NGS) platforms, scientists are now looking to apply these powerful molecular technologies to the vast pool of existing FFPE samples. The following are a handful of key ways in which NGS technologies can be applied to FFPE tissue:
Unfortunately, the fixation process also leads to extracted nucleic acids with poor quality, degradation, fragmentation, and deamination of cytosine bases, complicating transcriptomic-level investigations.2 “Formalin tends to crosslink the tissue to such an extent that it is hard to get fragments of DNA that are longer than, maybe, a hundred or two hundred base pairs,” says David Rimm, a pathologist at Yale University, who was interviewed by Nature in 2007 on this topic.1 The race is now on to generate methods for analyzing the degraded and modified biomolecules obtained from FFPE samples for molecular analysis.
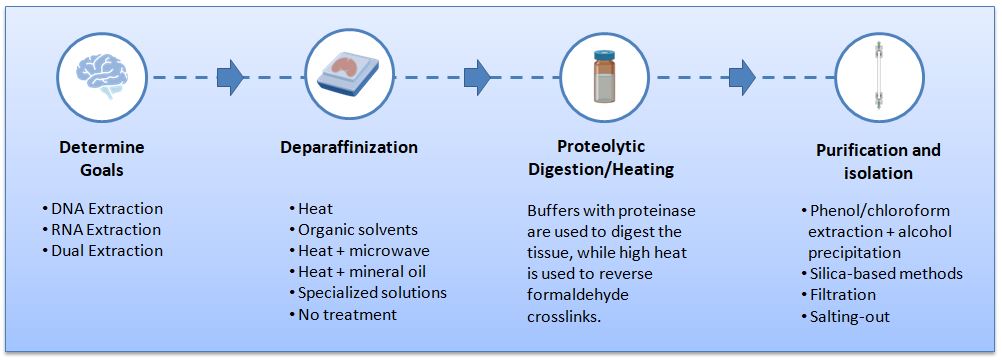
Figure 1. Procedures used to extract nucleic acids from formalin-fixed paraffin-embedded tissues.
Paraffin removal is often done with xylene or other organic solvents, but there are alternate methods such as direct melting with microwaves, using mineral oil and incubation at 90°C, or using specialized extraction reagents.3 Most researchers agree that the de-waxing step does not have significant effects on downstream nucleic acid analysis, though the toxicity of the deparaffinization reagent is worth considering.3
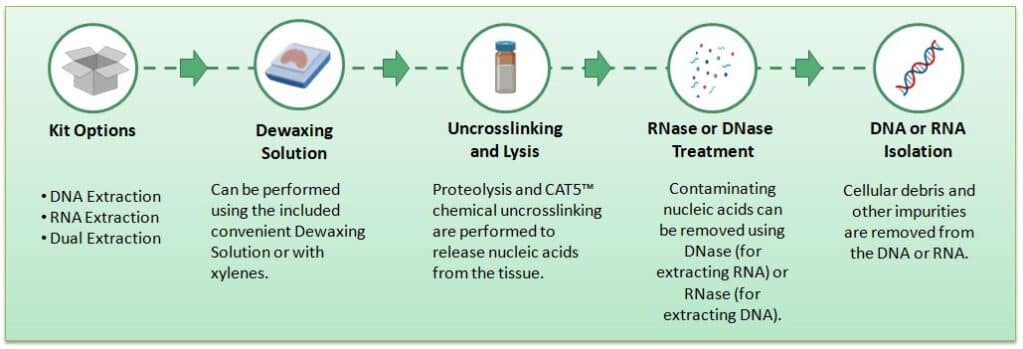
Figure 2. Workflow of DNAstorm™ 2.0 and RNAstorm™ 2.0 extraction from FFPE tissues.
Sanger sequencing, the first method for sequencing DNA, was first described in 1977 and involved sequencing a single base at a time. A little under 30 years later, modern NGS was developed and greatly reduced the time and cost of genome sequencing. This second technological leap was based on parallel sequencing of millions of relatively short DNA fragments and required reconstruction of the short reads when sequencing long stretches of DNA. Common sequencing platforms from manufacturers like Ion Torrent, Illumina, and GeneReader utilize this technology. While revolutionary, second-generation sequencing can be challenging, especially in the presence of structural variations and low-complexity regions. This was the impetus for third-generation sequencers (e.g., Pacific Biosciences and Oxford Nanopore Technology instruments), which can read lengths of more than 10 kb and are able to directly target unfragmented DNA molecules in real-time. Unfortunately, due to the fragmented and relatively poor quality of nucleic acids extracted from FFPE samples, long-read sequencing platforms are typically not compatible with data gleaned from FFPE tissue.5 Therefore, there remains the need for more effective methods for extracting genetic material extraction from FFPE tissue as well as downstream sequencing.
Optimizing the use of FFPE tissue with NGS promises great rewards in the form of access to a vast wealth of clinical and non-clinical data which can be used to study cellular processes in tissue. While challenges remain in extracting high-quality nucleic acids from FFPE tissue, recent advances in extraction methods offer hope for successful NGS analysis. Unlocking the genomic cache stored in FFPE tissue has the potential to revolutionize our understanding of diseases and pave the way for personalized medicine and scientific breakthroughs.

Julianne Davis earned an MSc in Behavioral Neuroscience from the University of Washington, Seattle, where she examined the role of memory in cost-based decision-making. She has also studied sensory integration at the Allen Institute and the neural basis of feeding, thirst, and metabolism at the University of California, San Francisco as a research scientist. Currently, she is a Technical Writer and Support Scientist at Biotium.