Aggregates of hyperphosphorylated Tau protein are linked to neurodegeneration and are a primary biomarker of Alzheimer’s Disease (AD). These tauopathies are often associated with synucleinopathies, including Parkinson’s Disease (PD), which are similarly characterized by the abnormal accumulation of aggregates of alpha-synuclein (α-Syn) protein in neural tissue. While protein aggregation that results in the formation of α-Syn and Tau deposits is a key event in these disorders, the primary neurotoxic effect seemingly originates from smaller oligomeric intermediates in the aggregation pathway. Therefore, oligomers have been proposed as novel drug targets or as biomarkers for disease detection. However, measuring oligomers in human biofluids is challenging as extreme sensitivity and specificity are required due to a vast excess of monomers.
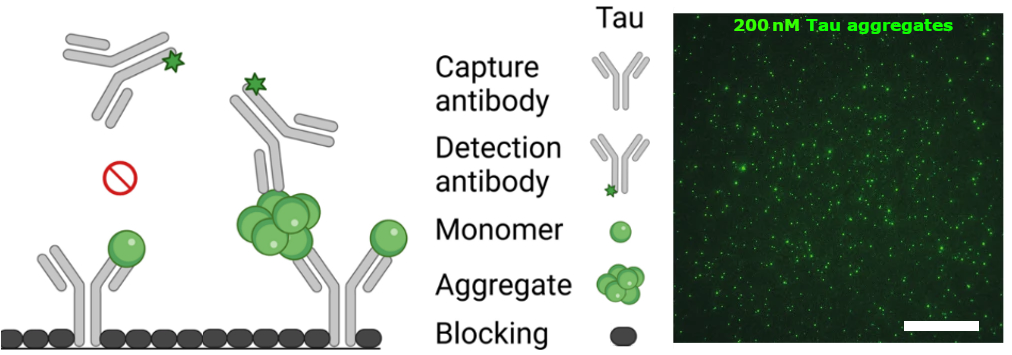
In a recent npj Parkinson’s Disease article, Blömeke et al. (2022) report a promising advancement towards oligomer-based diagnostics using a surface-based fluorescence intensity distribution analysis (sFIDA) protocol, a technology capable of single-particle sensitivity and specificity for protein aggregates. The authors demonstrate the clinical potential of this methodology by measuring α-Syn and Tau aggregate concentrations in 237 cerebrospinal fluid (CSF) samples from patients in five cohorts: PD, AD, dementia with Lewy bodies, progressive supranuclear palsy, and neurologically-normal controls. In this application of the sFIDA technique, antibodies directed against linear epitopes of α-Syn or Tau were immobilized on the surface of a microtiter plate. CSF samples containing both monomers and aggregates were then applied and allowed to bind to the capture antibodies. During the subsequent detection step, only bound aggregates were recognized by anti-Tau or anti-α-Syn monoclonal detection antibodies labeled respectively with Biotium’s CF®488A and CF®633 Dyes. This is possible because capture and detection antibodies that bind identical epitopes were used, and in the case of monomers the epitope was masked by the capture antibody. The assay surface was imaged by total internal reflection fluorescence (TIRF) microscopy, and single particles on the surface were quantified by image-data analysis. Using this technique, the authors showed that α-Syn and Tau aggregate levels successfully distinguished patients with different neurodegenerative diseases and that aggregate concentrations showed comparable discrimination to other conventional biomarkers: total Tau protein and phosphorylated Tau protein.
Learn more about Biotium’s high-performance CF® Dyes with exceptional brightness and photostability, Mix-n-Stain™ CF® Dye Antibody Labeling Kits, and a wide selection of other reagents for immunofluorescence microscopy.
Full Citation
Blömeke, L., et al. Quantitative detection of α-Synuclein and Tau oligomers and other aggregates by digital single particle counting. npj Parkinson’s Disease 8.1 1-13 (2022). https://doi.org/10.1038/s41531-022-00330-x

