PMA Real-Time PCR Bacterial Viability Kit – Legionella pneumophila (mip)
A kit designed to perform viability PCR on Legionella pneumophila. It contains the viability PCR dye PMA and primers to amplify the mip gene.
Please fill in the inquiry form and we will contact you shortly.
Wishlist updated! View wishlist
Product Description
This kit is designed to perform viability PCR on Legionella pneumophila. It contains the viability PCR dye PMA and primers to amplify the mip gene.
Features
- Selectively detect viable Legionella pneumophila using qPCR
- Contains primers to amplify the mip gene
- Works even in complex samples or mixed cultures
- Sufficient reagents to treat 80 bacterial cultures with PMA
- Sufficient reagents for 200 PCR reactions
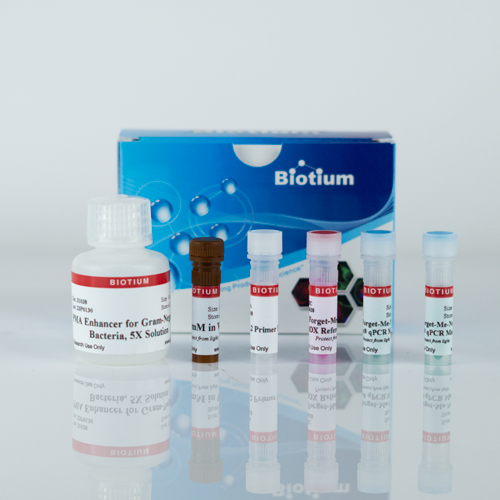
Kit components
- PMA dye
- PMA Enhancer for Gram Negative Bacteria
- Forget-Me-Not™ EvaGreen® qPCR Master Mix
- ROX reference dye
- mip primer mix, 5 uM
About this kit
Legionella pneumophila is a pathogenic species of gram-negative bacteria. It can infect the lungs and cause Legionnaire’s Disease. Legionella pneumophila is sometimes detected in water sources such as cooling towers and swimming pools. PCR to amplify the gene mip has been published and is highly specific for Legionella. The primers provided in the kit have been validated at Biotium for real-time qPCR using Forget-Me-Not™ EvaGreen® qPCR Master Mix.
To learn more about the advantages of determining bacterial viability using viability PCR, visit the Viability PCR Technology Page.
Strain-Specific Bacterial Viability PCR Kits
| Bacteria Strain | Gene name | Kit with PMA (catalog #) | Kit with PMAxx™ (catalog #) |
|---|---|---|---|
| Salmonella enterica | invA | 31033 | 31033-X |
| Mycobacterium tuberculosis | groEL2 | 31034 | N/A |
| Staphylococcus aureus | nuc | 31035 | N/A |
| MRSA | mecA | 31036 | N/A |
| E. coli O157:H7 | Z3276 | 31037 | 31037-X |
| E. coli | uidA | 31050 | 31050-X |
| Listeria monocytogenes | hly | 31051 | 31051-X |
| Legionella pneumophila | mip | 31053 | N/A |
Don’t see your favorite strain? Let us know at techsupport@biotium.com.
References
Download PMA and PMAxx™ References and a list of PMA and PMAxx™ Validated Bacterial Strains.