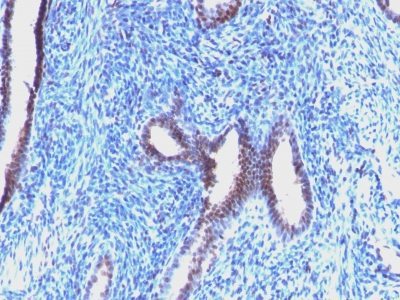
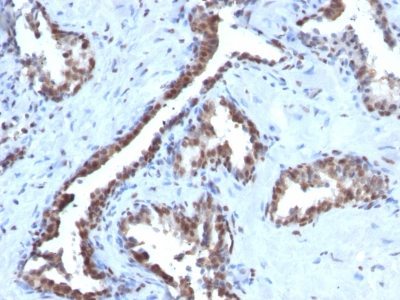
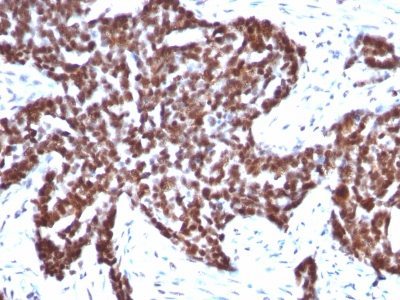
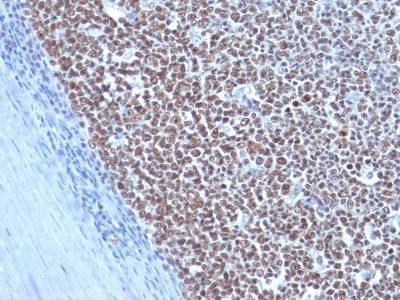
Cyclin B1 Monoclonal Mouse Antibody (CCNB1/1098)
Please fill in the inquiry form and we will contact you shortly.
Wishlist updated! View wishlist
Product Description
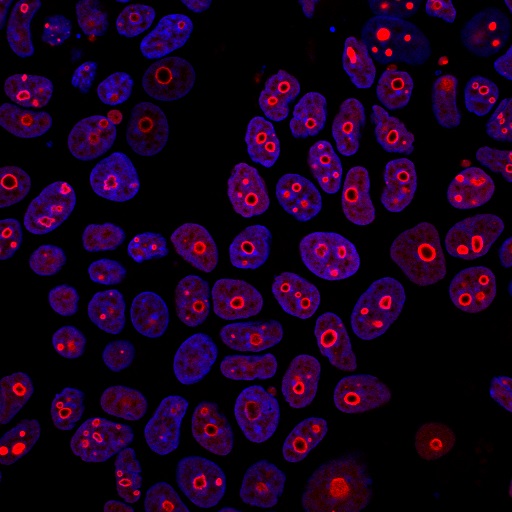
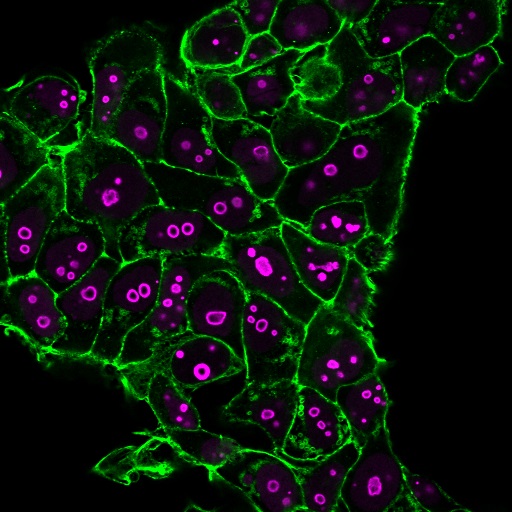
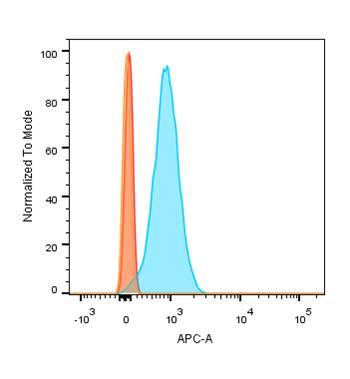
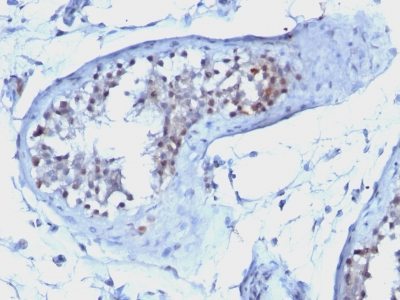
This antibody recognizes a protein of 55-62 kDa, identified as cyclin B1. In mammals, cyclin B associates with inactive p34cdc2, which facilitates phosphorylation of p34cdc2 at aa 14Thr and 15Tyr. This maintains the inactive state until the end of G2-phase. The inactive cyclin B-p34cdc2 complex continues to accumulate in the cytoplasm until the completion of DNA synthesis, when Cdc25, a specific protein phosphatase, dephosphorylates aa 14Thr and 15Tyr of p34cdc2 rendering the complex active at the G2/M boundary. This mitotic kinase complex remains active until the metaphase/anaphase transition when cyclin B is degraded. This degradation process is ubiquitin-dependent and is necessary for the cell to exit mitosis. So, cyclin B-p34cdc2 plays a critical role in G2 to M transition.
Primary antibodies are available purified, or with a selection of fluorescent CF® dyes and other labels. CF® dyes offer exceptional brightness and photostability. See the CF® Dye Brochure for more information. Note: Conjugates of blue fluorescent dyes like CF®405S and CF®405M are not recommended for detecting low abundance targets, because blue dyes have lower fluorescence and can give higher non-specific background than other dye colors.
Catalog number key for antibody number 1098, Anti-Cyclin B1 (CCNB1/1098)
| Antibody # prefix | Conjugation | Ex/Em (nm) | Laser line | Detection channel | Dye Features |
|---|---|---|---|---|---|
| BNC04 | CF®405S | 404/431 | 405 | DAPI (microscopy), AF405 | CF®405S Features |
| BNC88 | CF®488A | 490/515 | 488 | GFP, FITC | CF®488A Features |
| BNC68 | CF®568 | 562/583 | 532, 561 | RFP, TRITC | CF®568 Features |
| BNC94 | CF®594 | 593/614 | 561 | Texas Red® | CF®594 Features |
| BNC40 | CF®640R | 642/662 | 633-640 | Cy®5 | CF®640R Features |
| BNC47 | CF®647 | 650/665 | 633-640 | Cy®5 | CF®647 Features |
| BNC74 | CF®740 | 742/767 | 633-685 | 775/50 | CF®740 Features |
| BNCB | Biotin | N/A | N/A | N/A | |
| BNUB | Purified | N/A | N/A | N/A | |
| BNUM | Purified, BSA-free | N/A | N/A | N/A |