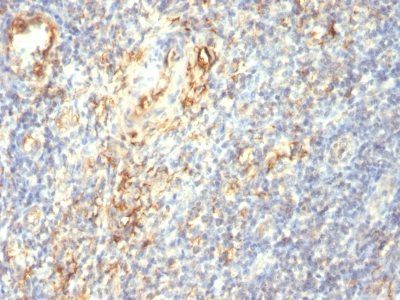
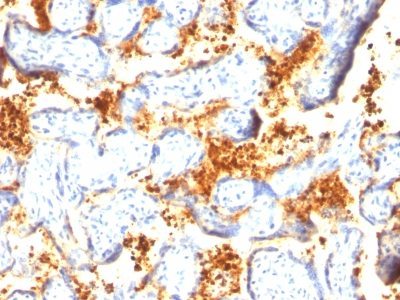
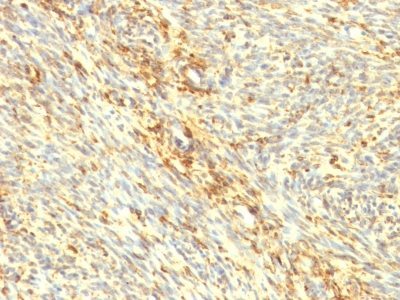
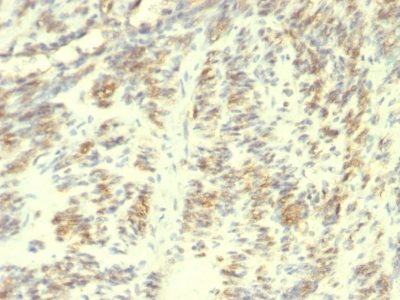
Transglutaminase-II Monoclonal Mouse Antibody (TGM2/419)
Please fill in the inquiry form and we will contact you shortly.
Wishlist updated! View wishlist
Product Description
Recognizes a 77-85 kDa protein, identified as cellular or tissue transglutaminase II (TGase II). Transglutaminases are enzymes that catalyze the crosslinking of proteins by epsilon gamma glutamyl lysine isopeptide bonds. While the primary structure of transglutaminases is not conserved, they all have the same amino acid sequence at their active sites and their activity is calcium-dependent. The protein encoded by this gene acts as a monomer, is induced by retinoic acid, and appears to be involved in apoptosis. Finally, the encoded protein is the autoantigen implicated in celiac disease. The identification of transglutaminase as the main antigen of endomysium antibodies allows a new diagnostic approach to celiac disease (CD), a genetic, immunologically mediated small bowel enteropathy that causes malabsorption. TGase II is implicated in programmed cell death, signal transduction, drug-resistance, cell growth, endocytosis, insulin secretion, cell adhesion, cataract formation, and wound healing.
Primary antibodies are available purified, or with a selection of fluorescent CF® dyes and other labels. CF® dyes offer exceptional brightness and photostability. See the CF® Dye Brochure for more information. Note: Conjugates of blue fluorescent dyes like CF®405S and CF®405M are not recommended for detecting low abundance targets, because blue dyes have lower fluorescence and can give higher non-specific background than other dye colors.
Catalog number key for antibody number 0419, Anti-Transglutaminase-II (TGM2/419)
| Antibody # prefix | Conjugation | Ex/Em (nm) | Laser line | Detection channel | Dye Features |
|---|---|---|---|---|---|
| BNC04 | CF®405S | 404/431 | 405 | DAPI (microscopy), AF405 | CF®405S Features |
| BNC88 | CF®488A | 490/515 | 488 | GFP, FITC | CF®488A Features |
| BNC68 | CF®568 | 562/583 | 532, 561 | RFP, TRITC | CF®568 Features |
| BNC94 | CF®594 | 593/614 | 561 | Texas Red® | CF®594 Features |
| BNC40 | CF®640R | 642/662 | 633-640 | Cy®5 | CF®640R Features |
| BNC47 | CF®647 | 650/665 | 633-640 | Cy®5 | CF®647 Features |
| BNC74 | CF®740 | 742/767 | 633-685 | 775/50 | CF®740 Features |
| BNCB | Biotin | N/A | N/A | N/A | |
| BNUB | Purified | N/A | N/A | N/A | |
| BNUM | Purified, BSA-free | N/A | N/A | N/A |
References
Yamanishi, K., et al. 1991. Molecular cloning of human epidermal transglutaminase cDNA from keratinocytes in culture. Biochem. Biophys. Res. Commun. 175: 906-913. |