As I pondered the title of this article, my thoughts immediately went back to a request by my PhD advisor to look at the effects on cell viability by an untested drug we suspect inhibits mitochondrial protein import. Cell viability you say? Where to begin…. The myriad of pathways, mechanisms, assays, and associated readouts to measure “viability” quickly made me realize just how comprehensive the term was. For those of you wondering how to get started with cell viability, let’s explore the many tools and metrics to help provide you with some direction.
Are there changes in metabolism or cellular activity?
Assays that measure cellular activity offer the most traditional readouts for viability (often directly called cell viability assays). These assays generally rely on determining relative levels of key metabolic co-factors or ATP. They are also commonly called cell proliferation assays, because they can be used to measure the relative number of live cells in different wells or samples.
Reductive potential
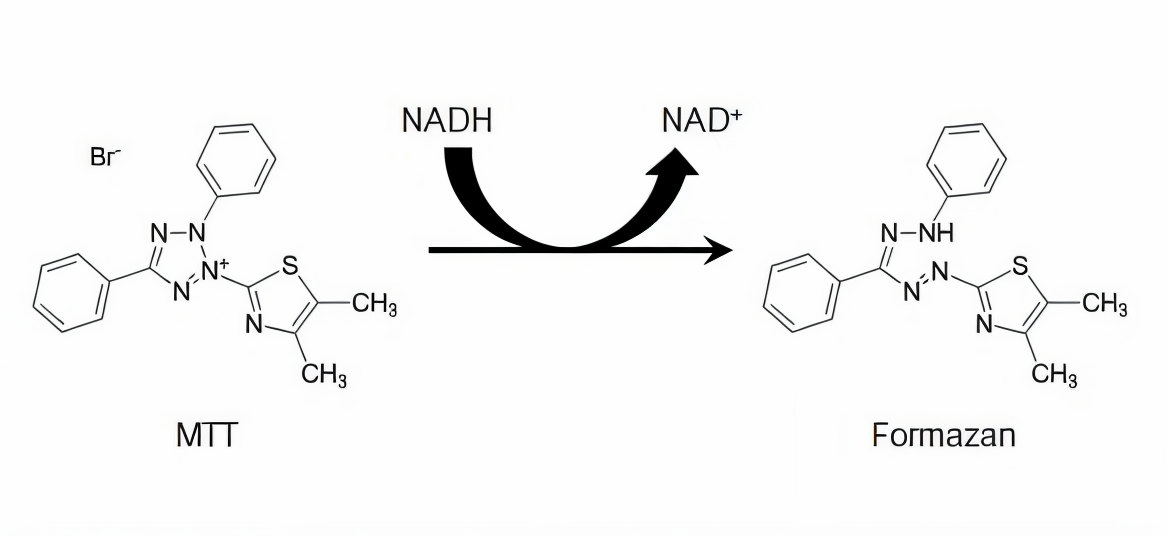
Reduction of MTT by NADH to form formazan product. From Riss et al. (2004). Reproduced under the Creative Commons license.
ATP levels
Another metric for cellular activity is the level of ATP, the molecule that provides the energy that drives cellular reactions, because only metabolically active cells can synthesize ATP. The most widely used method for measuring ATP is through a luminescent enzymatic assay that involves luciferase. This type of assay begins with a cell lysis step to release intracellular ATP. The ATP within the lysate is combined with luciferase and its substrate D-Luciferin. The reaction then generates luminescence which can be measured by a luminometer. Biotium’s Steady-ATP™ HTS Viability Assay Kit is a glow-type luminescence assay that functions this way and can be used for highly sensitive detection of ATP.
Cell proliferation assays
Cell viability can also be assessed by monitoring the cell proliferation rate of the culture. CFSE and ViaFluor® stains are membrane-permeant dyes that are non-fluorescent until they enter viable cells where they are hydrolyzed by cytoplasmic esterases to release fluorescent amine-reactive dyes. The dyes then covalently react with amine groups on intracellular proteins and are retained in the cell. Each subsequent cell division reduces the fluorescent signal by half, allowing each division to be monitored by flow cytometry.
Calcein-AM assays
Calcein-AM based assays are cell viability assays that require both intracellular esterase activity and intact plasma membranes (see cytotoxicity assays below). Similar to ViaFluor® Dyes, calcein-AM is non-fluorescent until it is hydrolyzed by intracellular esterases to release the green fluorescent dye calcein. However, cells require an intact plasma membrane to retain calcein fluorescence, and therefore only live cells will generate a signal. This enables calcein-AM to be used as a true endpoint viability assay and for cell killing assays.
Are my cells undergoing apoptosis?
Rather than measuring broad cellular activity, assays that are specific for apoptotic activity offer a more detailed understanding of cell viability and the mode of cell death. This category includes a variety of assays that measure various hallmarks of apoptotic cells.
Caspase activity
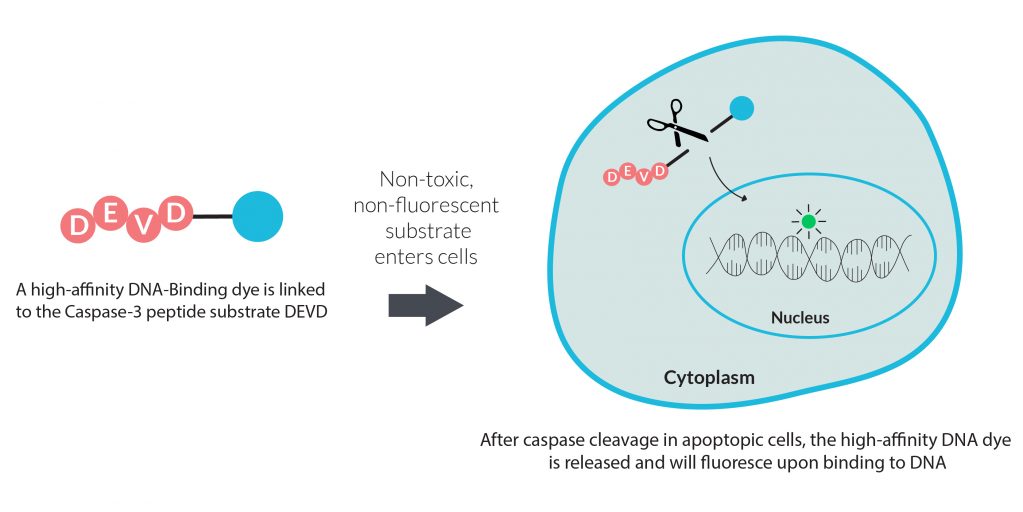
Measuring the activity of specific apoptotic proteases is a popular method for monitoring apoptosis. This type of assay is generally end-point and typically relies on a fluorogenic protease substrate that fluoresces after being cleaved by a specific protease. Biotium’s NucView® Caspase-3 Substrates are a similar type of assay. However, unlike other fluorogenic protease substrates, NucView® Caspase-3 Substrates release a high-affinity DNA binding dye that is retained in the nucleus. This allows real-time monitoring of both caspase-3 activity and changes to nuclear morphology.
Mitochondrial potential
Mitochondria aren’t simply the “Powerhouse of the Cell” we’ve come to know. They’re an extensive organelle network vital for essential cellular processes including metabolism, calcium signaling, apoptosis, and cell proliferation. Depolarization or loss of mitochondrial membrane potential is an early event in apoptosis. Consequently, assays monitoring mitochondrial potential are used as a key metric for assessing cell viability. These types of assays rely on cationic dyes that preferentially localize in the matrix of polarized mitochondria. Examples of cationic dyes include MitoView™ 633 as well as classic dyes like TMRM, TMRE, and JC-1. These types of dyes may also be used with NucView® for dual monitoring of caspase-3 activity and membrane potential in live cells.
Annexin V staining
Externalization of phosphatidylserine (PS) on the surface of the plasma membrane is a widely known indicator for apoptosis. Annexin V is a cellular protein that binds to PS and is commonly used as a probe for monitoring apoptosis. Annexin V can be used with dead cell dyes like propidium iodide (PI) or EthD-III for dual detection of apoptosis and necrosis, but can also be used with other cell viability probes. Biotium offers many CF® Dye Annexin V conjugates as well as a Dual Apoptosis Assay with NucView® 488 Caspase-3 Substrate & Annexin V and the Apoptosis and Necrosis Quantitation Kit Plus with CF®488A Annexin V and EthD-III.
TUNEL staining
Internucleosomal dsDNA breaks are another key indicator of apoptosis. Apoptosis-induced DNA breaks in fixed cells and tissues can be detected using the TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling) assay. This assay involves a TdT enzyme that catalyzes the addition of labeled dUTP to the 3’-ends of DNA double-strand breaks. The labeled cells can then be detected with microscopy or flow cytometry.
Are my cells dead? Cytotoxicity or Membrane Integrity Assays
An alternative way to determine the number of live cells in a population is to measure the number of dead cells. Because the loss of membrane integrity is a key indicator for cell death, cytotoxicity assays (aka live/dead assays) rely on membrane-impermeant dyes or substrates to be excluded outside of living cells. The widely known trypan blue dye that’s used for cell counting functions this way. Several of Biotium’s cell viability products also rely on membrane integrity to segment live and dead cell populations. This includes Live-or-Dye™ stains as well as PI, EthD-III, TOTO®-1, TO-PRO®-1, and other dead cell specific nucleic acid stains. PMA and PMAxx™ dyes also rely on membrane integrity to determine cell viability but are mainly used for viability PCR in microorganisms.
Other questions to ask
Real-time or end-point?
Another important consideration is determining whether your study requires only a simple yes or no answer to cell viability, or requires consistent measurements in real-time. Cell viability assays that involve cell lysis (such as Steady-ATP™ or MTT assay) or require fixation before staining (such as TUNEL) are end-point and not suitable for real-time measurements. Other cell viability assays that are non-toxic and live cell-compatible such as NucView®, Annexin V, or dead cell nucleic acid stains are suitable for either end-point or real-time analysis.
To fix or not to fix?
You may need to evaluate methods depending on whether your samples are already fixed, or if you will need to fix cells after staining for further analysis, like immunofluorescence staining. Most viability or apoptosis assays require cells to be alive during the assay. Some assays like NucView® and Live-or-Dye™ can withstand fixation and permeabilization after staining is complete. Others like dead cell nucleic acid stains, MitoView™, or calcein-AM must be assayed in live cells and cannot be fixed. On the other hand, TUNEL staining is used to detect apoptosis in cells or tissue sections that are fixed before the assay is done.
Are you planning on doing high-throughput screening (HTS)?
Careful consideration of the assay workflow is needed when screening large libraries of mutants, compounds, or other conditions for affects on cell viability. End-point assays where measurements aren’t time sensitive are generally more suitable for high-throughput screening (HTS) because the samples can be done in multi-well plates. This includes our Caspase-3 DEVD-R110 Fluorometric HTS Assay Kit and reductive potential assays. However, automated microscopy screening platforms have been developed that can accommodate high-throughput imaging and detection of cellular phenotypes (Boutros et al., 2015). For example, NucView® Caspase substrates have been validated with Essen/Sartorius Incucyte® and Molecular Devices ImageXpress® imaging platforms for high-content monitoring of apoptosis (Aftab et al., 2014; Antczak et al., 2009). In addition, assays that require multiple wash or incubation steps such as TUNEL, calcein-AM, or Live-or-Dye™ staining may require more complex high-throughput screening workflows compared to homogeneous assays that can be directly added to the cell culture medium. Examples of single-step homogeneous assays include XTT, resazurin, NucView®, MitoView™, and dead cell DNA dyes.
Do you have the right equipment?
Before planning your cell viability experiment, you should ensure that you have the proper equipment to detect the relevant outputs. Assays where the signal fills the entire cell or is released into the medium can be used with standard microplate readers for fluorescence (calcein-AM, resazurin), or absorbance (MTT, XTT). Flash-type assays where sample measurement is time sensitive, a plate reader with injectors may be required. Other assays where the signal is localized to specific compartments within the cells require a specialized imaging microplate reader, fluorescence microscope, or flow cytometer. For time-lapse imaging a real-time imaging system is also required.
Is your assay validated for your model system?
There is considerable variance in cell physiology between different model organisms and experimental conditions. Consequently, consistent and reproducible results in your model system are critical to validating any conclusions on cell viability. When designing your cell viability experiment, it is important to validate the assay procedure in your model system as well as establish the appropriate positive and negative controls.
Conclusions
Understanding what data is needed to reach a noteworthy conclusion on cell viability is crucial to selecting the right type of assay. Hopefully, this article provides some insight on the mechanisms behind the various types of cell viability assays to help make that decision. Learn more about Biotium’s full selection of cell viability and apoptosis reagents, as well as stains and probes for a variety of cellular structures.
References
- Riss, T. L., Moravec, R. A., Niles, A. L., Duellman, S., Benink, H. A., Worzella, T. J., & Minor, L. (2004). Cell Viability Assays. In Assay Guidance Manual. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/23805433
- Boutros, M., Heigwer, F., & Laufer, C. (2015, December 3). Microscopy-Based High-Content Screening. Cell, Vol. 163, pp. 1314–1325. https://doi.org/10.1016/j.cell.2015.11.007
- Aftab, O., Nazir, M., Fryknäs, M., Hammerling, U., Larsson, R., & Gustafsson, M. G. (2014). Label free high throughput screening for apoptosis inducing chemicals using time-lapse microscopy signal processing. Apoptosis, 19(9), 1411–1418. https://doi.org/10.1007/s10495-014-1009-9
- Antczak, C., Takagi, T., Ramirez, C. N., Radu, C., & Djaballah, H. (2009). Live-cell imaging of caspase activation for high-content screening. Journal of Biomolecular Screening, 14(8), 956–969. https://doi.org/10.1177/1087057109343207